Maximising the effectiveness of therapeutic vaccines a step closer
Scientists at UCL have significantly boosted the effectiveness of a therapeutic vaccine designed to control chronic hepatitis B (HBV) infection, after uncovering and thwarting 'the enemy within'.
Published in Science Translational Medicine, researchers say the findings in mice are a breakthrough in the field of vaccine development, as they reveal how to enhance the antiviral T cell response that therapeutic vaccines are intended to trigger. This has been a long-time problem and puzzle for immunologists around the world.
While there is an effective prophylactic (preventative) HBV vaccine to prevent new cases, this study focused on how to enhance the response to therapeutic vaccines (those administered after a disease or infection has already occurred) among the estimated 240 million people who are already chronically infected.
Chronic HBV is the most common cause of liver cancer in the world, and each year globally, the infection causes an estimated 880,000 deaths from liver cirrhosis and hepatocellular carcinoma/liver cancer (HCC).
For the study, mice with chronic HBV were vaccinated with ChAdOx1-HBV, Vaccitech, Oxford, which is currently being tested in Phase II trials in humans.
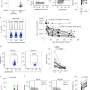
Researchers found that removing natural killer (NK) cells, a white blood cell that normally fights infection, boosted the response of the antiviral CD8+T cells induced by the therapeutic vaccination and enhanced the control of HBV.
Co-lead author, Dr. Mariana Diniz (UCL Division of Infection & Immunity), said: "Our study shows that natural killer cells living in the liver, rather than helping T cells to fight the virus after vaccination, instead suppress them and prevent them from controlling the infection."
Building on this, the team discovered that NK cells use the PD-1 inhibitory pathway to suppress T cells. Further, when researchers activated the NK cells with cytokines (proteins that stimulate the immune system) and blocked the PD-1 pathway, NK cells were converted them from 'suppressors' into 'helpers', boosting the number of vaccine-induced T cells able to control HBV.
Using blood and liver patient samples, the research team went on to show that the findings also applied to hepatitis B patient samples in vitro, reinforcing the potential to translate this discovery into new and improved human vaccines.
Co-lead author, Professor Mala Maini (UCL Division of Infection & Immunity), said: "NK cells are increasingly recognised to have potent positive or negative regulatory capacities in addition to their direct antiviral and anti-tumour functions.
"What we found happening in the HBV-infected liver is like one battalion of soldiers—the natural killer cells—turning on a fellow battalion—the T cells—rather than the common enemy, hepatitis B.
"Our findings describe an immunotherapeutic combination that can overcome this constraint to boost the response to therapeutic vaccination in hepatitis B, an approach that is potentially also applicable to liver cancer."
The vaccine used in the study, ChAdOx1-HBV, Vaccitech, Oxford, contains HBV antigens delivered by a chimpanzee adenoviral vector, using the same backbone platform as the AstraZeneca (ChAdOx1 nCoV-19) SARS-CoV-2 vaccine.
Importance to therapeutic vaccine development
Professor Maini added: "Restoring or rescuing an effective T cell response is the primary goal of many immunotherapies being developed for chronic viral infections and malignancies.
"To date, therapeutic vaccines have shown limited success but remain a key strategy to promote immune responses targeting specific antigens.
"Our study uncovers a new mechanism that regulates T cell responses to therapeutic vaccines and an immunotherapeutic approach to overcome this; insights which could be applied to develop more effective treatments."
Study limitations
This study was performed using a mouse model for HBV infection that does not fully reproduce the natural infection process observed in humans. However, the model resembles the state of low HBV replication seen in several human carriers and mimics the exhausted immunological state of immune cells in chronic HBV infection and is, therefore, useful to study therapeutic interventions. Findings from the mouse model were reproduced using patient samples.
The research was conducted with collaborators at the University of Oxford.
More information: Erin Gatza et al, Manipulating the Bioenergetics of Alloreactive T Cells Causes Their Selective Apoptosis and Arrests Graft-Versus-Host Disease, Science Translational Medicine (2011). DOI: 10.1126/scitranslmed.3001975