This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
How the body's defenses keep their weapons in check
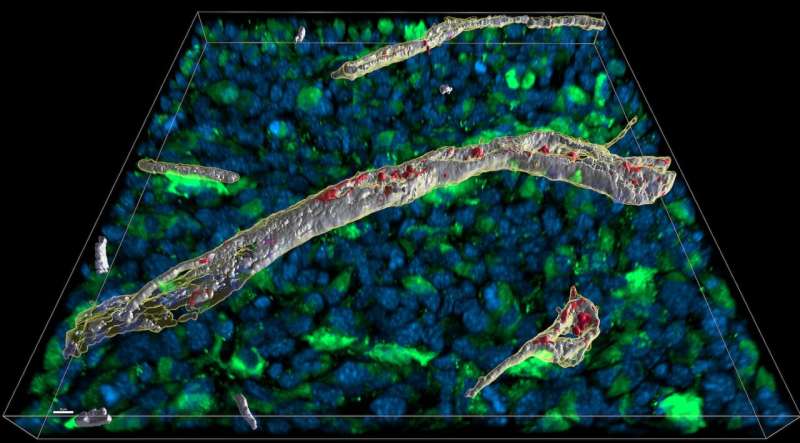
The signaling molecules of the immune system should trigger a response only where necessary. To prevent a life-threatening spread to the rest of the body, connective tissue can absorb these molecules like a sponge. A team led by Thomas Blankenstein presents this mechanism in Nature Immunology.
When the T cells of the immune system communicate, they do so with the help of cytokines. An important member of the cytokine family is interferon-gamma—a protein that activates the body's defenses, particularly in the fight against viruses and bacteria.
Over the course of evolution, the human body has developed a variety of strategies to prevent the immune response from overshooting its mark. Another important mechanism has now been discovered by a German-French research team led by Professor Thomas Blankenstein, head of the Molecular Immunology and Gene Therapy Lab at Berlin's Max Delbrück Center.
It all hangs on just four amino acids
In a paper published in the journal Nature Immunology, the scientists explain how interferon-gamma uses four amino acids to bind to the extracellular matrix of connective tissue, which forms a web between individual cells and thus mediates intercellular contact. The study's first author, Dr. Josephine Kemna, explains that this binding prevents the cytokine from spreading throughout the entire body and triggering dangerous immune responses.
When the amino acids required for binding are lacking, she says, the result is a serious impairment of the body's defenses. Kemna was a member of Blankenstein's team from 2017 to 2022. Last year, she moved to the Berlin biotech company T-knife Therapeutics—a spin-off from Blankenstein's lab. Kemna completed her doctorate with this latest study, in which Charité—Universitätsmedizin Berlin also played a key role. The research was supported by a grant from the Wilhelm Sander Foundation.
The starting point for the study was an observation made by Blankenstein and his team a few years ago: "We noticed that the molecular structure of the cytokine interferon-gamma differs greatly from species to species," explains Dr. Thomas Kammertöns, another member of the team who also works at the Institute for Immunology at Charité.
He supervised Kemna's doctoral thesis together with Blankenstein and is listed as last author. "However, one short sequence of four amino acids, known as the KRKR motif, has remained practically unchanged over the entire evolution of vertebrates—i.e., over 450 million years—in all 50 species we studied." Based on this finding, the team deduced that the KRKR motif must play an important role in the function of the cytokine—and decided to test this hypothesis.
Quickly turning toxic in the blood
The researchers started out using a mouse model developed by Kammertöns, which allowed them to regulate the concentration of interferon-gamma that was produced. "We were already able to determine from this model that interferon-gamma becomes toxic very quickly, and that animals with high concentrations of this signaling molecule in their blood fall ill within a few days," explains Kammertöns.
Biochemical analyses also revealed that once the protein is secreted via the T cells with its four positively charged amino acids, it binds to the negatively charged extracellular matrix—namely, to the molecule heparan sulfate.
"This ensures that interferon-gamma is retained locally, and prevents it spreading throughout the body," says Kammertöns. However, given that the structure of heparan sulfate differs depending on the tissue, cell type or even cell state, the ability of connective tissue to bind interferon-gamma can also vary, adds Professor Hugues Lortat-Jacob of the Université Grenoble-Alpes, who was also involved in the study.
In the next step, the group turned to Dr. Ralf Kühn, head of the Genome Editing & Disease Models Lab at the Max Delbrück Center, to help develop a model that would produce interferon molecules without a KRKR motif. To do this, Kühn and his team removed the four amino acids from the cytokine in mice using the CRISPR-Cas9 gene editing technique.
"For a long time, scientists have believed that the signaling molecule is dependent on this binding site to function at all," Kammertöns says. "So we first had to prove that this is not the case." And the team was indeed able to show that, even without the KRKR motif, interferon-gamma still attaches to its receptor on the surface of cells and performs its usual role in the immune response.
Highly potent defense mechanisms
Usually, the immune system would then fight the viral infection and eventually eliminate it. However, for the mice lacking the four amino acids in their interferon-gamma, that was not the case. "The animals' immune systems were still able to regulate immune responses for viruses that elicit only very brief inflammatory reactions," Kammertöns reports, saying that in these cases, the amount of interferon-gamma in the blood did initially increase but then fell again very quickly.
"Yet when the mice were infected with LCM viruses, which cause a flu-like disease called lymphocytic choriomeningitis and keep the immune system busy for a longer period of time, the gene-edited mice quickly became ill due to the high concentrations of interferon-gamma in their blood."
"In my view, it is clear from our research that our immune system has developed highly potent mechanisms to keep its own defenses in check," says first author Kemna. If these mechanisms fail to work properly, she says, the immune system can end up damaging its own organism due to the toxic effect of certain molecules as they continue to spread.
"The mechanism we have uncovered shows that evolution has ensured toxic molecules generally act only where they are needed—that is, where the T cell recognizes a virus-infected cell."
Protection against deadly infection
"This study is of fundamental importance for immunology and our understanding of many inflammatory diseases in the human body," says Kammertöns. He also explains that the extracellular matrix has a different structure in males and females, so the newly discovered mechanism could explain why some infectious and autoimmune diseases progress so differently in men and women.
"We would never have made these new findings without the outstanding collaboration with our French colleague Hugues Lortat-Jacob, who has been researching extracellular matrices for more than 30 years and is one of the world's leading experts in this field," Kammertöns adds.
Kammertöns is now planning the next phase of the study with his group leader Blankenstein and scientists at University Medical Center Freiburg. Together, they are going to test their latest findings on a new model. "We want to work with so-called wildlings—mice that have already undergone several infections and whose immune systems therefore elicit a response more similar to that of a human," Kammertöns says.
"Over the course of its evolution, the immune system has developed increasingly powerful weapons in a sort of arms race against pathogens," summarizes Blankenstein. "Our work has uncovered a new mechanism that acts a counter balance to this arsenal of weapons without reducing the efficiency of the immune response—just four amino acids in interferon-gamma prevent infectious diseases from causing many more deaths."
It therefore makes sense going forward to gain a better understanding of the exact details of the interaction between interferon-gamma and the extracellular matrix.
More information: Josephine Kemna et al, IFNγ binding to extracellular matrix prevents fatal systemic toxicity, Nature Immunology (2023). DOI: 10.1038/s41590-023-01420-5