This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Prolonging the survival of patients with gastric cancer
Gastric cancer is the fifth most commonly diagnosed malignancy worldwide. It ranks a sad third in cancer-related causes of death. The reason for this is late diagnosis coupled with rapid spread of tumor cells in the body.
In an international clinical study co-chaired by investigators from Leipzig University's Faculty of Medicine, researchers investigated a substance that is expected to prolong the survival of patients and also established a new clinically relevant biomarker. The results have been published in The Lancet.
"The results of the study are important for cancer research. They show that patients with gastric cancer who have been treated with zolbetuximab live longer. The progression of the tumor disease is delayed and overall survival of patients is improved. We expect that the study will lead to the approval of this drug in Europe and as a result also in Germany. This is an important step for those affected by this serious and often fatal cancer," said Professor Florian Lordick, director of the University Cancer Center Leipzig. The experienced oncologist helped to design the recently published study at the international level and ensured that German patients were able to participate.
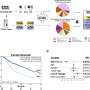
In this study with participants from 20 countries, a total of 565 patients were treated with zolbetuximab in combination with chemotherapy or a placebo and chemotherapy. The test substance was developed in Germany more than ten years ago and has since been thoroughly researched through all study phases until it was ready for clinical use in patients.
In the current study, treatment with zolbetuximab and chemotherapy significantly reduced the risk of disease progression or death by 25 percent compared to patients treated with chemotherapy alone. The aim of the study was therefore achieved. The most common adverse events among participants were nausea, vomiting and decreased appetite, but overall the side effects of the combination of zolbetuximab and chemotherapy proved to be manageable.
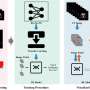
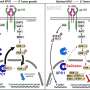
Furthermore, the study establishes a new clinically relevant biomarker in gastric cancer called claudin-18 isoform 2 (CLDN18.2). This biomarker can be identified using a staining method in tumor biopsies, a procedure to remove cells or tissue from an affected area. If 75 percent of the tumor cells stain positive for the membrane protein CLDN18.2, the drug zolbetuximab is likely to work particularly well.
Zolbetuximab is given as an intravenous infusion to patients with advanced gastric cancer in combination with chemotherapy. It then docks via the bloodstream to the tumor cells carrying CLDN18.2, where it triggers an immune response that contributes to the death of the tumor cells.
"Following the introduction of trastuzumab ten years ago, this is the second targeted antibody that can prolong the survival of patients with advanced gastric cancer, and I have been privileged to play a key role in its clinical development," said Professor Lordick.
More information: Kohei Shitara et al, Zolbetuximab plus mFOLFOX6 in patients with CLDN18.2-positive, HER2-negative, untreated, locally advanced unresectable or metastatic gastric or gastro-oesophageal junction adenocarcinoma (SPOTLIGHT): a multicentre, randomised, double-blind, phase 3 trial, The Lancet (2023). DOI: 10.1016/S0140-6736(23)00620-7