This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Scientists discover how anti-viral cytokines can blunt the immune response to tuberculosis
A new collaborative study recently published in the journal Cellular Immunology by researchers from Trinity's School of Medicine and the Trinity College Institute of Neuroscience (TCIN) is closing the research gap on the actions of an important protein of the immune system—type 1 interferon, whose actions remain poorly understood.
Type one interferons are cytokines responsible for killing viruses, including COVID-19. Cytokines are small proteins that are crucial in controlling the growth and activity of other immune and blood cells. When released, they signal the immune system to do its job.
Although cytokines play a positive, protective role, many patients suffer from diseases where chronic production of type 1 interferons make them sick. These include persons with the auto-immune disease systemic lupus erythematosus (SLE) and people suffering with chronic tuberculosis (TB).
It is not known though how type one 1 interferons impair the ability of the immune system to fight bacterial infection.
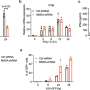
Dr. Gina Leisching, Senior Research Fellow, Clinical Medicine, School of Medicine has led the group to study the influence of type 1 interferons on the immune system, using an animal model. This study demonstrates that type 1 interferon treatment induces an inflammatory state characterized by the increased production of white cells, and inflammatory intermediate metabolites as well as immune cell metabolic rewiring—which interferes with the ability of macrophages to fight bacteria.
Specifically, the group have identified type one interferons as impairing the metabolic response to Mycobacterium tuberculosis, the bacteria that causes TB.
Dr. Leisching said, "This work has provided new evidence that chronic type I interferons blunt the immune response and explains why patients with diseases that are driven by these immune proteins are prone to infections.
"We now have new targets to test to see if we can reverse this poor response by either boosting immune cell function or limiting the effects of type I interferons. We are now working with immune cells from SLE patients to gain more insight into whether these effects are observed in all immune cells or only a select few."
These findings now identify type 1 interferons as a potential target for the development of host directed therapies in patients who suffer from excess production of this cytokine.
This approach is already used in treating SLE—an autoimmune disease in which the immune system attacks its own tissues, causing widespread inflammation and tissue damage in the affected organs—but might now be looked at in pre-clinical models of tuberculosis. The field of TB needs new adjuvant therapies that support the host, especially as antibiotics are rendered redundant by the increasing development of resistance in the Mycobacterium tuberculosis bacteria (XDR TB).
Anjali Yennemadi, Ph.D. candidate, Clinical Medicine, School of Medicine, said, "We are very excited by these findings as they help to close a gap in the literature by showing what a significant effect type I interferons have on altering immune cell metabolism. This is particularly interesting to us as we are now working with SLE patients to understand why they are more susceptible to infections, including TB.
"Going forward, with the use of more advanced experimental and computational techniques, we hope to uncover potential diagnostic and/or therapeutic targets specific to chronic production of type I interferons in SLE and TB."
More information: Anjali S. Yennemadi et al, Chronic IFNα treatment induces leukopoiesis, increased plasma succinate and immune cell metabolic rewiring, Cellular Immunology (2023). DOI: 10.1016/j.cellimm.2023.104741