This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
reputable news agency
proofread
A new treatment option for a form of sudden hair loss

A new type of medication, JAK inhibitors, can effectively treat moderate to severe alopecia areata, a hair loss condition that has been historically hard to treat.
A study of its effectiveness, by Dr. Brett King and Dr. Brittany Craiglow of Yale University, was published in August in a supplement to the Journal of the American Academy of Dermatology.
"Because alopecia areata is an inflammatory condition, a JAK inhibitor will essentially reduce the inflammation that is fueling the disease and bring your immune system back into balance," said dermatologist Dr. Sandra Johnson. She is an adjunct professor at the University of Arkansas for Medical Sciences, in Little Rock, who was not involved with the study.
"The development of JAK inhibitors has given us another treatment to improve the lives of patients with alopecia areata," Johnson said in a news release from the American Academy of Dermatology.
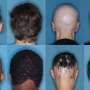
The condition is more common in kids but can happen at any age. It involves sudden hair loss with affected patches that grow larger. In some cases, it spreads to the entire head or body.
It is also more common in those who have a close blood relative with the disease and in people who have been treated for cancer with a drug called nivolumab (Opdivo). Medical conditions such as asthma, hay fever, eczema, thyroid disease, vitiligo and Down syndrome also increase alopecia areata risk.
Courtney Martens, a mother of two from Edmond, Okla., participated in a clinical trial at Johnson's practice in Fort Smith, Ark.
Martens began living with alopecia areata at 38, when she first noticed a bald patch about the size of a silver dollar on her scalp. Eventually, Martens lost all hair on her scalp, eyebrows and eyelashes.
She was diagnosed with alopecia areata in 2017. Her immune system was causing her body to attack its own hair follicles.
"It's pretty traumatizing," Martens said in the news release. "Most people think it's just hair, but it was exhausting because it became what everybody talked about. It was like I lost my identity because I was always the girl with pretty hair growing up."
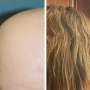
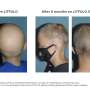
After she received treatment with a JAK inhibitor at Johnson's practice, Martens was able to completely regrow her hair. She said that she feels blessed that the treatment worked for her.
The new study credits JAK inhibitors with ushering in a new era, making treatment of moderate-to-severe alopecia areata possible. The authors noted that two drugs—baricitinib and ritlecitinib—are approved, and a third, deuruxolitinib, is moving toward approval. Clinical trials are also ongoing.
Diagnosis of alopecia areata involves an examination of the area of hair loss as well as a person's nails. Blood tests may be needed to rule out other diseases caused by the immune system.
Besides JAK inhibitors, contact immunotherapy can be used to change a person's immune system so that it stops attacking their hair follicles. Other treatments options include a disease-modifying antirheumatic medication called methotrexate and anti-inflammatory corticosteroids.
"We now have more treatment options than ever before for alopecia areata patients, and they are providing results for people for whom previous treatments were not effective," Johnson said. "It's important to know that no one treatment works for everyone. Your board-certified dermatologist can recommend the treatment options that work best for you."
More information: The U.S. National Institutes of Health has more on alopecia areata.
Brett A. King et al, Janus kinase inhibitors for alopecia areata, Journal of the American Academy of Dermatology (2023). DOI: 10.1016/j.jaad.2023.05.049
2023 HealthDay. All rights reserved.