This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Discovery of mechanism involved in dental inflammation paves way to therapies against bone loss

By means of experiments in mice, researchers have made discoveries about the mechanisms involved in dental pulp inflammation and root apex damage that pave the way for a search for medications that can be used to inhibit the bone loss due to endodontic infection (in dental pulp, the soft tissue inside teeth).
The key element in the mechanisms in question is tumor necrosis factor-α receptor-1, TNFR1, which becomes pro-inflammatory on binding to the cytokine TNF-α.
In one of the situations analyzed, the TNF-α-TNFR1 signaling pathway protected teeth and permitted repair, but in the other case, it acted in the opposite direction, leading to an inflammatory process with bone loss. The difference was due to the presence or absence of microorganisms.
One of the studies, reported in the Journal of Endodontics, showed that TNFR1 is involved in the formation of reparative dentin after dental pulp capping, a procedure in which bioactive material is placed over exposed pulp to encourage healing at the site of injury.
When TNFR1 is ablated (genetically removed or deactivated), the inflammatory response is altered and expression of key mineralization proteins (dentin sialoprotein and osteopontin) is inhibited, leading to dental pulp necrosis and development of apical periodontitis. In this manner, the researchers demonstrated in vivo that this pro-inflammatory signaling pathway is important to cell differentiation and synthesis of proteins that control the process of dental biomineralization, a crucial process for successful dental healing.
In the other study, reported in the same journal, the scientists observed that the TNF-α-TNFR1 signaling pathway is responsible for mediating bone tissue degradation after root canal contamination. The pathway plays an important role in inflammation and bone loss when the root canal is contaminated by microorganisms. If it is blocked, the negative effects can be reduced.
"Our group has researched the biological mediators involved in stem cell differentiation and biomineralization of teeth and bones. We study periapical lesions, a very specific type of lesion involving the penetration of bacteria into the dental root canal and resulting in contamination of the canal," Francisco Wanderley Garcia de Paula-Silva, last author of both articles, told Agência FAPESP. Paula-Silva is a professor at the University of São Paulo's Ribeirão Preto Dental School (FORP-USP) in Brazil.
The articles resulted from the Ph.D. research of Luciano Aparecido de Almeida Júnior in pediatric dentistry at FORP-USP, with Paula-Silva as his thesis advisor.
Paula-Silva and his group recently published an article written for and reviewed by children to explain the tooth regeneration process, emphasizing the cells and molecules involved.
They also have a YouTube channel with several films on oral health "literacy".
Cavities (also called caries) are the main oral health problem among children and adolescents, according to preliminary data from the National Oral Health Survey now being conducted by the Brazilian government. Other findings show that 45% of older people aged 65-74 need immediate treatment of some kind owing to dental pain or infection and that 48% of adults in the 35-44 age group need at least one elective dental procedure.
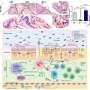
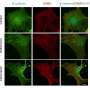
In the study on reparative dentin formation, the researchers compared the dental pulp repair response of TNFR1-deficient mice with that of wild-type mice (C57BL/6 strain) as the control group. Pulp capping was performed with mineral trioxide aggregate. After 7 and 70 days, tissues were collected and assessed by histological analysis and immunohistochemistry.
The conclusion was that genetic ablation of TNFR1 modified the inflammatory process and inhibited expression of mineralization proteins, leading to dental pulp necrosis and development of apical periodontitis.
In the study of the role of the TNF-α-TNFR1 signaling pathway, the researchers induced apical periodontitis by inoculating oral microorganisms into the molar root canals of mice lacking TNFR1 and compared their response with that of the wild-type control group after 7, 14, 28, and 42 days. They performed microcomputed tomography analysis, with the participation of FORP-USP's Endodontics Research Laboratory, as well as histopathological and PCR evaluation, among others.
The conclusion was that lower recruitment of neutrophils in TNFR1-deficient mice at 14, 28 and 42 days resulted in reduced apical periodontitis area and volume at 42 days. The number of osteoclasts was also lower in TNFR1-deficient mice at 14 and 42 days.
Osteoclasts are cells that mediate bone loss in pathological conditions via demineralization and degradation. They make space for osteoblasts to create new bone tissue in areas that are growing or need repair.
Paula-Silva and his group are now looking for therapeutic approaches based on the results of these two studies. "We realized from these research findings that a treatment protocol could be developed if this inflammatory response is blocked by inhibiting the receptor. We're still in the initial stages, based on an animal model, but it's a very interesting target," he said.
More information: Luciano Aparecido de Almeida-Junior et al, Reparative Dentin Formation Following Dental Pulp Capping is Mediated by TNFR1 In Vivo, Journal of Endodontics (2023). DOI: 10.1016/j.joen.2023.06.015
Luciano Aparecido Almeida-Junior et al, TNF-α–TNFR1 Signaling Mediates Inflammation and Bone Resorption in Apical Periodontitis, Journal of Endodontics (2023). DOI: 10.1016/j.joen.2023.07.013