This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Investigational drug restores parathyroid function in rare disease
An investigational drug, encaleret, restored calcium levels in people with autosomal dominant hypocalcemia type 1 (ADH1), a rare genetic disorder marked by an imbalance of calcium in the blood and urine, as well as abnormally low levels of parathyroid hormone, which regulates blood calcium levels.
Led by clinician-scientists from the National Institute of Dental and Craniofacial Research (NIDCR) at the National Institutes of Health's Clinical Center, results from the clinical trial are published in the New England Journal of Medicine.
In the mid-phase clinical trial, 13 participants with ADH1 received oral doses of the investigational drug for about 24 weeks. By the end of the trial, the treatment restored every participant's blood calcium level to normal, and urine calcium approached normal levels. Levels of parathyroid hormone also normalized.
"It was amazing to see that every participant responded to the treatment. In literally minutes after taking the medication orally, the levels of parathyroid hormone increased dramatically," said senior author and NIDCR endocrinologist Michael Collins, M.D.
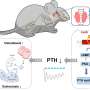
Our bodies need calcium for proper functioning of many organs and tissues, including the bones, teeth, heart, muscles, and nerves. However, in people with ADH1, calcium levels in the blood are unusually low, leading to symptoms that can range from tingling limbs, muscle cramps, and brain fog to life-threatening seizures. Between 1.4 and 3.9 in 100,000 people in the U.S. are affected by the disease. While current therapies can help manage symptoms, there are no approved treatments that address the root cause of the condition.
"Conventional therapy is to raise the blood calcium level with calcium supplements and activated vitamin D," said principal investigator and NIDCR pediatric endocrinologist Rachel Gafni, M.D. "However, too much of an increase could cause kidney stones or damage kidney tissues, leading to kidney failure in worst-case scenarios. The patients need better treatments, so we're not constantly walking on a tightrope."
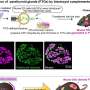
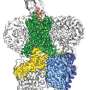
Encaleret is thought to exert its effects by acting on faulty calcium-sensing receptors that are peppered throughout the kidneys and pea-sized organs in the neck called parathyroid glands. In healthy people, these receptors act like thermostats to monitor and control calcium levels. However, in patients with ADH1, the receptors are too sensitive and falsely interpret normal blood calcium levels as high. As a result, the parathyroid glands do not make enough parathyroid hormone and the kidneys flush too much calcium out of the body. This leads to low calcium levels in the blood and high levels in the urine.
"This clinical trial, which was made possible by a public-private partnership, really demonstrates the value of collaboration and the institute's commitment and success in translating research into health," said NIDCR Director Rena D'Souza, D.D.S., Ph.D.
Encaleret appeared to be safe and caused no serious side effects. However, because parathyroid hormone raises blood calcium levels in part by drawing calcium from the bones, the researchers noted a need to assess the treatment's long-term effects on the skeleton. The researchers also speculate encaleret may have broader implications. An ongoing clinical trial led by Dr. Collins and NIDCR staff clinician Iris Hartley, M.D., is testing whether the treatment can help correct calcium levels in people whose parathyroid glands have been damaged by surgery.
"Because ADH1 can be passed on to offspring, the participants are not just doing this for themselves; it's for their children, grandchildren, and great-grandchildren," said Dr. Gafni. "One day, hopefully, we'll be writing prescriptions for an effective treatment thanks to them."
More information: Rachel I. Gafni et al, Efficacy and Safety of Encaleret in Autosomal Dominant Hypocalcemia Type 1, New England Journal of Medicine (2023). DOI: 10.1056/NEJMc2302708