This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New molecule to fight against transthyretin amyloidosis
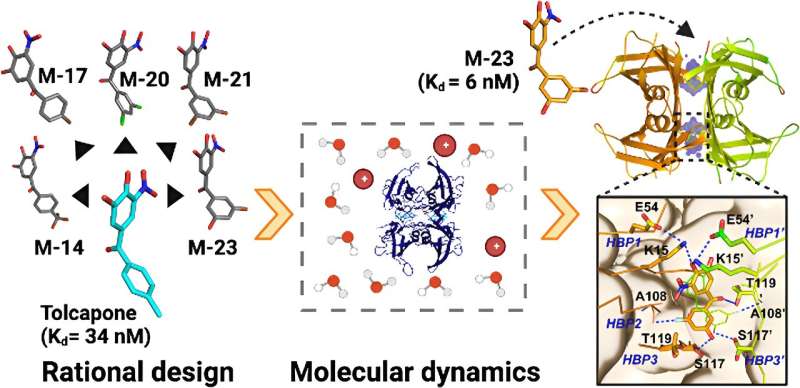
Researchers from IBB-UAB have developed a second-generation molecule that significantly improves the stabilization and inhibition of the aggregation of proteins involved in Transthyretin Amyloidosis (ATTR), a group of minority degenerative diseases.
The new molecule, called PITB (Pharmacokinetically Improved TTR Binder), is the result of a series of studies aimed at improving both the activity and pharmacokinetic profile of Tolcapone, a drug that the same group of researchers proposed in 2016 for the treatment of ATTRs and which is currently already in Phase 2 clinical trials.
ATTRs are a group of diseases caused by the accumulation of amyloid deposits of the transthyretin (TTR) protein. When TTR becomes unstable due to genetic mutations or aging, deposits form and can accumulate in various organs of the body, mainly in the heart and peripheral nerves.
In two studies recently published in the Journal of Medicinal Chemistry and the European Journal of Medicinal Chemistry, researchers describe the design and synthesis of PITB and how its structure binds with much stronger affinity and greater selectivity than Tolcapone to TTR, both to the wild-type protein and to the two most common and clinically relevant mutations, such as those associated with familial amyloid polyneuropathy and familial amyloid cardiomyopathy.
The new compound significantly stabilizes and inhibits TTR aggregation showing enhanced stabilizing activity in human plasma, especially in individuals affected by familial amyloid polyneuropathy. In addition, researchers note that PITB exhibits excellent pharmacokinetics and oral bioavailability, with no evidence of hepatotoxicity in cell culture.
Salvador Ventura, professor of Biochemistry and Molecular Biology and head of the Protein Structure and Aggregation Group at IBB, points out that "PITB has all the desired characteristics to be one of the most effective drugs in the fight against ATTR, and at a more affordable price than any current alternative."
The researchers now hope to secure new funding to conduct in vivo efficacy and toxicity testing, as well as formulation studies. In addition, they are contemplating the creation of a spin-off company to move towards the first clinical trials, which would be carried out at Son Llàtzer and Vall d'Hebron hospitals.
More information: Francisca Pinheiro et al, Development of a Highly Potent Transthyretin Amyloidogenesis Inhibitor: Design, Synthesis, and Evaluation, Journal of Medicinal Chemistry (2022). DOI: 10.1021/acs.jmedchem.2c01195
Francisca Pinheiro et al, PITB: A high affinity transthyretin aggregation inhibitor with optimal pharmacokinetic properties, European Journal of Medicinal Chemistry (2023). DOI: 10.1016/j.ejmech.2023.115837