This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Research finds non-small cell lung cancer drug lorlatinib targets additional protein
Targeted cancer drugs are widely used because of their ability to inhibit specific proteins involved in cancer development with fewer side effects compared to chemotherapy drugs. But targeted therapies can often inhibit other unknown proteins. These hidden targets may also contribute to the drug's anticancer effects and potentially offer a path for the drug to be repurposed for other cancers controlled by the hidden target.
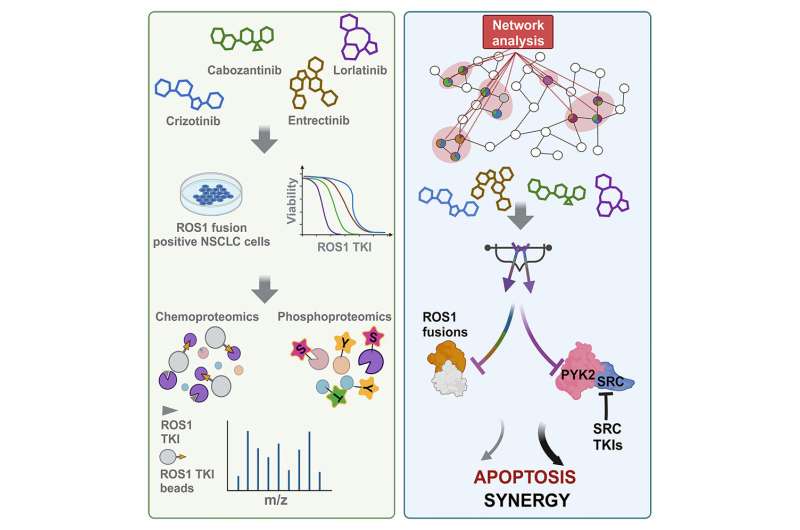
In a new study published in Cell Chemical Biology, Moffitt Cancer Center researchers demonstrate this, showing that the ROS1 inhibitor lorlatinib has activity against an additional protein called PYK2. The team also reveals the mechanisms of this inhibition.
"Drugs with polypharmacology mechanisms, meaning agents that target multiple functionally relevant proteins, may have the potential to effectively overcome drug resistance, improve anti-cancer activity of single-targeted inhibitors and produce a more durable patient response and remission," said Uwe Rix, Ph.D., lead study author and senior member of the Department of Drug Discovery at Moffitt.
Rix and a team of Moffitt researchers wanted to determine whether cancer drugs that target the protein ROS1 in non-small cell lung cancer have additional hidden targets. They performed their studies in cancer models that were driven by the cancer promoting properties of the oncogenic ROS1 protein; therefore, they needed to develop an investigative platform that would be able to identify any potential additional targets that might be hidden by the dominant properties of ROS1.
They first compared the activity of a panel of different ROS1 inhibitors and discovered that the drug lorlatinib had the highest inhibitory activity against ROS1 but also disproportionately high cell viability when compared to other ROS1 inhibitors. These observations suggested that lorlatinib may target additional proteins that contribute to its anticancer effects.
The team performed additional laboratory experiments and found that lorlatinib also targets the protein PYK2. This protein plays an important role in focal adhesions, which are cell structures that are involved in several processes including cell adhesion, movement and survival. Their studies also showed that the protein SRC physically associated with PYK2 and ROS1 and helped control the activity of PYK2. These data suggested that inhibition of ROS1, PYK2 and SRC in combination would have greater anticancer effects than either agent alone.
Further studies confirmed that drug combinations targeting each protein resulted in enhanced cancer cell death and reduced cell survival than either agent alone. Likewise, lorlatinib combined with an SRC inhibitor also resulted in enhanced anticancer effects.
The researchers hope these results and their investigative platform will lead to a better understanding of the mechanisms of action of lorlatinib and the development of drug combinations with improved clinical activity.
More information: Yi Liao et al, Differential network analysis of ROS1 inhibitors reveals lorlatinib polypharmacology through co-targeting PYK2, Cell Chemical Biology (2023). DOI: 10.1016/j.chembiol.2023.09.011


















