This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers design potential therapy to prevent brain deterioration in children with rare genetic conditions

A research team at the Francis Crick Institute and Great Ormond Street Hospital (GOSH)/UCL Great Ormond Street Institute of Child Health have identified new potential treatments for children with rare genetic conditions of blood vessels, which cause severe, lifelong, and disabling symptoms like seizures and impaired development.
Through two papers published in the Journal of Investigative Dermatology, the researchers showed that problems with calcium underlie these diseases. Stabilizing calcium levels in the brain could therefore be a target for new treatments, to protect the brain from damage over time.
"Mosaic mutations"—genetic changes which appear in some cells in the body but not all—in two genes, GNAQ or GNA11, cause a spectrum of conditions including Sturge-Weber syndrome and Phakomatosis Pigmentovascularis with Dermal Melanocytosis (PPV-DM).
These mutations affect the skin, brain and eyes, leading to seizures, impaired development, severe headaches, blindness, and birthmarks on the face. Although children are born with the conditions, the brain symptoms get progressively worse over the first years of life. People can have a normal life expectancy, but many will experience serious and disabling symptoms throughout their lives.
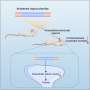
Calcium deposits were known to build up in the brain in these conditions. They form parallel lines in the blood vessels, called "tramlining," which can be seen on X-rays. Doctors believed the calcium deposits were non-specific signs of brain damage, but this research challenges this view.
The researchers examined 42 children with either Sturge-Weber syndrome or PPV-DM at GOSH. They found that 74% of the children had at least one abnormal measurement of calcium in the blood, and their calcium brain deposits were getting worse over time. Surgery for epilepsy in five patients revealed calcium deposits inside brain cells and tiny blood vessels in the brain.
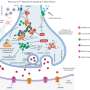
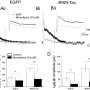
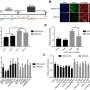
The researchers then studied patient samples in the lab at the Crick, as well as blood vessel cells engineered with and without the mutations. This approach clarified exactly what was happening in the cells with the mutations, identifying that too much calcium was being released inside cells all the time. This effect was so strong that it led to the cells pulling in even more calcium from outside to keep up with the demand, through transporters on the cell called CRAC channels. The team concluded that this chronic calcium imbalance could underpin the clinical symptoms.
To try and treat the cells in the lab, the team tested a new genetic therapy to silence the overactive gene and a drug to block the CRAC channels. Both treatments were able to improve the calcium problems, but the genetic therapy was the most effective.
The researchers hope early intervention with the genetic therapy or with a drug that targets the calcium pathway will prevent the calcium deposits building up in the brain cells and contributing to seizures.
Veronica Kinsler, Principal Group Leader of the Mosaicism and Precision Medicine Laboratory at the Crick, Professor of Pediatric Dermatology and Dermatogenetics at GOSH/UCL, and NIHR Research Professor, said, "It's always such a shock for the family if a child is born with Sturge-Weber syndrome or PPV-DM as these rare conditions aren't picked up on ultrasound screening, and they don't run in families so they can happen to anyone.
"By using the power of science to work out exactly what is going on in this disease, we have finally been able to understand the calcium deposits in the brain that doctors have seen in the clinic for years. With the support of all the patients who have participated and the funders, we've been able to use this new insight to design potential treatments. We think there's a window of opportunity where we can now try to make a difference for children diagnosed with these severe diseases."
Antonella Perini, president of the Associazione Sindrome di Sturge Weber Italia, said, "For our organization, which gathers together patients affected by a rare disease and their families, scientific research stands as our paramount ally and hope. Thus, a research project yielding such significant and potentially impactful results fills us with satisfaction and a sense of confidence in the future."
More information: Nicole Knöpfel et al, GNAQ/GNA11 mosaicism is associated with abnormal serum calcium indices and microvascular neurocalcification, Journal of Investigative Dermatology (2023) DOI: 10.1016/j.jid.2023.09.008
Zecchin, D. et al, GNAQ/GNA11 mosaicism causes aberrant calcium signalling susceptible to targeted therapeutics., Journal of Investigative Dermatology (2023). DOI: 10.1016/j.jid.2023.08.028.