This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Identifying new potential in cancer-killing T cells
When Joanina Gicobi began her Ph.D. degree five years ago at Mayo Clinic Graduate School of Biomedical Sciences, she was interested in thinking creatively about ways to improve cancer treatment. In particular, she was curious about the body's natural ability to fight tumors: Could that process be improved? In the laboratory of Haidong Dong, M.D., Ph.D., she joined efforts to learn more about the T cells that are part of an inherent cancer-killing immune response.
"From the beginning, she was asking impressive questions about an issue that's been a great challenge," says Dr. Dong, whose lab focuses on cancer immunology and immunotherapy.
The inquisitive approach to problem-solving paid off. Dr. Gicobi, who recently completed her Ph.D. thesis, Dr. Dong and a team of Mayo Clinic researchers have provided a new understanding of the body's cancer-fighting T cells, known as CD8+ T cells.
In an article published in Science Advances, the research team describes a previously unknown type of CD8+ T cell that can withstand the toxicity of treatments like chemotherapy and radiation therapy. The cells, which they call resilient T cells, remain functional, suggesting there may be additional opportunity after chemotherapy and radiotherapy to target the cancer via the immune system. What's more, the team identified a potential strategy to bolster the cancer-killing capabilities of these hardy cells.
Scrutinizing cells that survive
One detail that has baffled researchers about CD8+ T cells is their persistence, particularly in patients who seem to have run out of treatment options.
Some people with advanced cancer undergo what's known as "salvage therapy" to ease late-stage symptoms. Salvage therapy may include chemotherapy and radiotherapy to shrink tumors that are not responding to immunotherapy or are too large to remove surgically, though the treatments are typically not expected to be a cure.
However, researchers had noticed that even after several rounds of therapies that kill off immune cells along with the cancer, some patients sometimes still had cancer-killing CD8+ T cells circulating in their blood. And those cells, however beaten down, seemed capable of rallying an immune response.
"But we didn't know the nature of these particular CD8+ T cells, how to identify them or what might make them unique," Dr. Dong says, adding that the lab hit numerous dead ends as they tried to characterize the cells. "It was not a straightforward route as we searched for answers."
The team monitored a cohort of patients who underwent salvage radiotherapy and then examined those patients' T cells. People who responded better to radiotherapy had T cells that used cellular energy most efficiently and could still kill cancer cells. Somehow the cells were less "exhausted" than other T cells. However, the phenotype didn't fully distinguish what was special about the cells.
Following the data
In her research, Dr. Gicobi set out to look for molecular markers particular to the T cells that rebound after cancer treatment. Using RNA sequencing, which can provide a map of all potential gene expression in a cell, she came across interesting findings that pointed back to certain genes. Dr. Gicobi noted several genes that were slightly different from those in typical CD8+ T cells.
Among them, she found a gene related to the cells' metabolic pathway, the process by which the cells produce and burn energy. This aligned with the earlier finding, and Dr. Gicobi felt encouraged to delve into the metabolism of the cells as a key differentiator. "Dr. Dong has told me through my training, 'Always follow the data,'" says Dr. Gicobi.
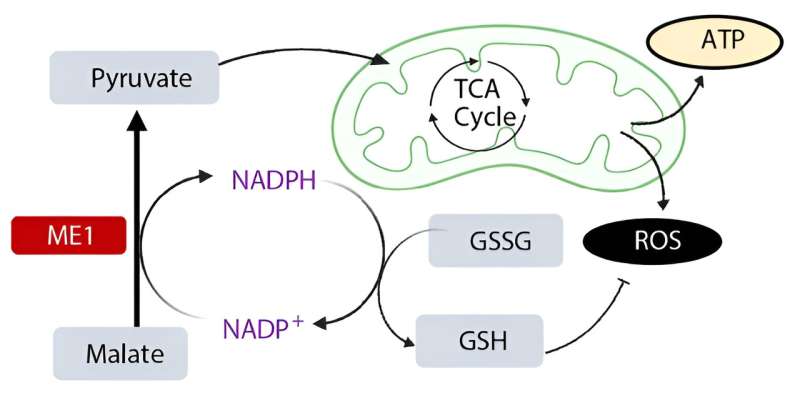
As the team members looked further, they found the cells had heightened expression of the enzyme called malic enzyme 1, or ME1. "That was really interesting to us because ME1 is known to deal with molecules called reactive oxygen species, a metabolic byproduct that is important for the functions of immune cells but toxic when in excess. So we were looking at an enzyme that helps reduce the negative effects of metabolic pathways," she says. The finding helped confirm that the resilient T cells in people are a distinct subgroup of CD8+ T cells.
Then, Dr. Gicobi had the idea that tinkering with ME1 to address the function and energy of immune cells might heighten their response to a tumor.
She engineered the resilient T cells to overproduce ME1. The laboratory experiments found the increase of ME1 revived the cells and made them even better at killing off tumor cells. Importantly, Dr. Gicobi says, "We were able to increase their cancer-killing capacity without the accumulation of reactive oxygen species that induce cellular exhaustion."
During her graduate training, Dr. Gicobi presented the findings at the annual meeting of the American Association of Immunologists.
Says Dr. Dong, "The excitement and the questions from attendees were a sign that this work suggests new approaches and more options for improving T cell health, which is an important avenue for the field."
He says further research is necessary to determine how resilient T cells affect patient outcomes and why some people have resilient T cells and others don't. He also hopes to learn more about the mechanisms underlying the function of resilient T cells and to explore the potential development of ME1 as a therapy.
But an important first step has been the understanding that the presence of resilient T cells indicates the perseverance of the body's inherent immune response. "This is one milestone toward new discoveries that can hopefully be translated to help patients," he says.
More information: Joanina K. Gicobi et al, Salvage therapy expands highly cytotoxic and metabolically fit resilient CD8 + T cells via ME1 up-regulation, Science Advances (2023). DOI: 10.1126/sciadv.adi2414