This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
New monoclonal antibody for treating cerebral ischemia injury

A research team from the LKS Faculty of Medicine at the University of Hong Kong (HKUMed) has successfully developed a neutralizing monoclonal antibody, 6H2, which targets a harmful protein known to exacerbate damage caused by ischemic stroke.
Through pre-clinical experiments on animals, the team found that the 6H2 antibody effectively attenuates ischemic stroke injury, offering potential for a highly effective pharmacological therapy for patients with ischemic stroke.
This breakthrough is the result of the concerted efforts of research teams from HKUMed's Department of Pharmacology and Pharmacy, the State Key Laboratory of Pharmaceutical Biotechnology, and the Department of Medicine, School of Clinical Medicine, HKUMed, as well as the Shenzhen Institute of Advanced Technology, Chinese Academy of Sciences.
The findings were published in the Journal of British Pharmacology, and patent applications for the newly developed 6H2 antibody were filed in China, Europe and the United States.
Stroke is a major cause of permanent disability and death worldwide. In Hong Kong, 13,500–14,000 individuals suffer from stroke every year, 80% of whom are diagnosed with ischemic stroke, caused by blocked brain blood vessels. Currently, there are limited treatment options; tissue plasminogen activator (tPA) is the sole FDA-approved drug. However, tPA has potential side effects, like brain bleeding, and must be administered promptly. Novel therapies are therefore urgently needed.
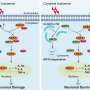
The harmful protein that worsens ischemic stroke is adipocyte fatty acid binding protein (A-FABP). HKUMed researchers discovered that A-FABP disrupts the blood-brain barrier by inducing matrix metalloproteinase 9 (MMP-9), worsening the effects of stroke. Higher circulating A-FABP levels were observed in mice with middle cerebral artery occlusion (MCAO) surgery and stroke patients, implicating it as a potential therapeutic target.
A previous study of the HKUMed research team found that an A-FABP inhibitor, BMS309403, showed promise but other studies demonstrated potential side effects on the heart. Therefore, a specific and potent monoclonal antibody to neutralize the protein may be a better therapeutic strategy for ischemic stroke.
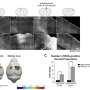
The researchers evaluated the impact of circulating A-FABP in ischemic stroke by conducting experiments on A-FABP-deficient mice. The mice were replenished with synthetic A-FABP (rA-FABP) into the bloodstream and were then subjected to MCAO surgery to simulate a stroke. The replenishment of rA-FABP worsened stroke outcomes, resulting in larger infarct size, brain swelling, more severe neurological deficits, and a lower survival rate. These findings highlight the detrimental effect of circulating A-FABP in ischemic stroke outcome.
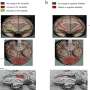
Using the mouse hybridoma technique, the research team generated monoclonal antibodies targeting A-FABP. The monoclonal antibody 6H2 demonstrated strong binding to A-FABP in both humans and mice. 6H2 can effectively block harmful A-FABP effects, including certain protein activation and MMP-9 production in macrophages, which can cause inflammation and damage. The efficacy and safety of 6H2 were further evaluated in both mice with simulated strokes and healthy mice.
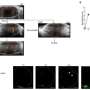
In wild-type mice with MCAO surgery, 6H2 treatment significantly alleviated blood-brain barrier disruption, reduced brain swelling, decreased infarction size, improved neurological function, and lowered mortality risk. These positive effects were observed by a reduction in cytokines and MMP-9 production, known contributors to inflammation and tissue damage.
Long-term 6H2 treatment had no obvious adverse effects in healthy mice, confirming its safety. These findings establish circulating A-FABP as a promising target for ischemic stroke treatment, validating 6H2 as a potential monoclonal antibody drug candidate.
Based on these findings, "our team is now collaborating with the Shenzhen Institute of Advanced Technology, Chinese Academy of Sciences to identify the structural determinant of 6H2 action and advance its humanization," said Professor Ruby Hoo Lai-chong, Assistant Professor, Department of Pharmacology and Pharmacy, HKUMed.
"With the pre-clinical evaluation of the humanized neutralizing monoclonal antibody on the horizon, we are optimistic that these efforts will lay the foundation for a revolutionary approach to the treatment of ischemic stroke," added Professor Hoo.
More information: Boya Liao et al, Development of a therapeutic monoclonal antibody against circulating adipocyte fatty acid binding protein to treat ischaemic stroke, British Journal of Pharmacology (2023). DOI: 10.1111/bph.16282