This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Combining CAR-T cells and inhibitor drugs for high-risk neuroblastoma
Chimeric antigen receptor (CAR)-T cell therapy is a potent emerging weapon against cancer, altering patients' T cells so they can better find and destroy tumor cells. But CAR-T cell therapy doesn't work well in every cancer—including many cases of neuroblastoma, a cancer that begins in young children's nerve tissue and can metastasize to multiple areas of the body.
Neuroblastoma can be deadly: Children with high-risk neuroblastoma have a five-year survival rate of only 50%. Roberto Chiarle, MD, at Boston Children's Hospital and his colleagues now report a way to make CAR-T cell therapy work better in neuroblastoma. They hope to begin testing it soon in children at high risk.
Giving CAR-T cells more targets
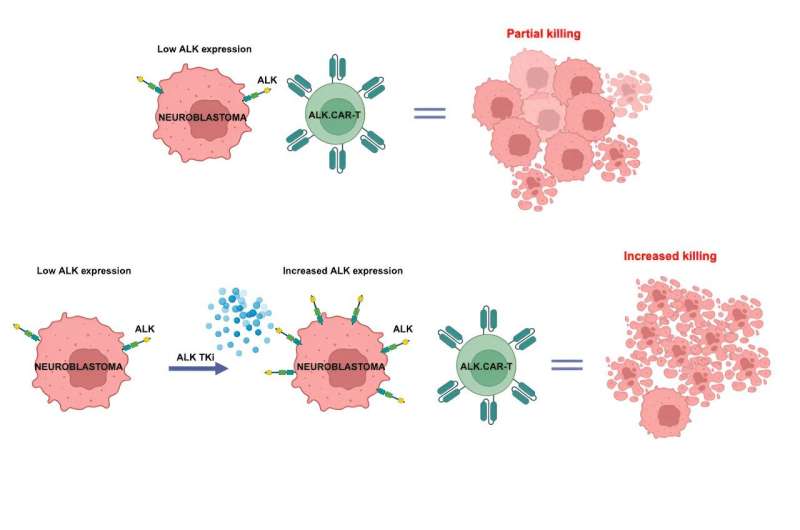
Chiarle and colleagues had previously developed CAR-T cells that specifically target the ALK receptor, a known oncogene that drives many cancers. However, not every patient with neuroblastoma has high enough levels of ALK receptors on their tumor cells to attract a strong CAR-T cell attack.
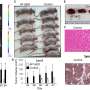
Led by research fellow Elisa Bergaggio, Ph.D., Chiarle's team tried adding an ALK inhibitor drug to the CAR-T cell treatment. They found that the inhibitor not only silences oncogenic signaling by ALK receptors, but it also increases numbers of these receptors on the cell surface—presenting more targets for CAR-T cells in patients with low ALK expression.
"The ALK receptors are present but inactivated by the ALK inhibitor, and because they're not active, they stay on the cell surface," explains Chiarle. "This increased surface expression facilitates CAR-T cell engagement and killing activity."
Curbing metastatic neuroblastoma
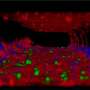
As the team reports in Cancer Cell, the combination of ALK-targeted CAR-T cells and ALK inhibitors proved to be a potent therapy in mice with metastatic neuroblastoma. Mice receiving the combination had significantly reduced tumor growth and better survival.
Chiarle and Bergaggio are now working to create an enhanced, more potent 2.0 version of the therapy. In the meantime, Dana-Farber/Boston Children's Cancer and Blood Disorders Center has filed for permission from the Food and Drug Administration to test the combination therapy in children with refractory or relapsed neuroblastoma. Susanne Baumeister, MD, of Dana-Farber/Boston Children's will run the clinical trial, hoped to open this spring.
Children with relapsed or refractory neuroblastoma would first receive escalating doses of ALK-targeting CAR-T cells alone. If this is shown to be safe, the ALK inhibitor would be added. CAR-T cells for each patient will be manufactured in the gene therapy facility at Dana-Farber Cancer Institute.
"Relapsed high-risk neuroblastoma remains a significant clinical problem," says Suzanne Shusterman, MD, who runs the Neuroblastoma Program at Dana-Farber/Boston Children's and will be a co-investigator on the trial. "Dr. Chiarle's preclinical data are very encouraging and we are very excited to have this therapy available to our patients."
More information: Elisa Bergaggio et al, ALK inhibitors increase ALK expression and sensitize neuroblastoma cells to ALK.CAR-T cells, Cancer Cell (2023). DOI: 10.1016/j.ccell.2023.11.004