This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers identify immune dysfunction as a possible aspect of polycystic ovary syndrome pathology
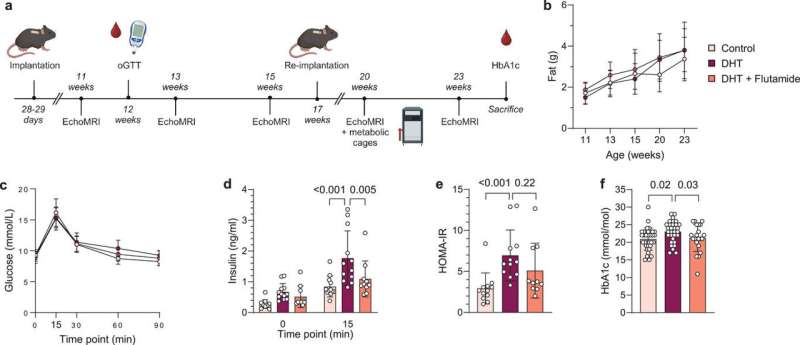
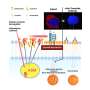
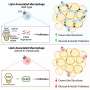
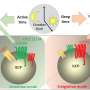
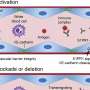
A new study shows that hyperandrogenism—a key characteristic of polycystic ovary syndrome (PCOS)—affects immune cell populations in reproductive, metabolic and immunological tissues in a PCOS-like mouse model.
These findings are of great importance as it is known that immune dysfunction is an essential part of reproductive complications and metabolic disease, which are very common among women with PCOS.
However, as the study shows that hyperandrogenism affects different tissues in unique ways, any possible treatments would have to be carefully tailored to target specific tissue dysfunctions. Since concurrent treatment with an androgen receptor antagonist prevented many changes in the mouse model used in the study, combination therapies that include both anti-androgens and other drugs that target specific altered immune pathways could be explored.
The study is published in the journal Advanced Science.
One of the most fascinating findings was the clear alterations of immune cells in adipose tissue, despite an unaltered fat mass of the androgen exposed mice. It is well known that immune cells in adipose tissue contribute to insulin resistance in overweight and obese individuals, but here the researchers produced an insulin resistant mouse model that mimics normal weight women with PCOS.
The impact of androgens on immune cells in adipose tissue is therefore very interesting considering the high prevalence of insulin resistance and type-2 diabetes among normal weight women with PCOS.
Another rather surprising finding was the drastic decrease of eosinophils in the uterus, as these don't express the androgen receptor. This shows that androgens play a broader and more complex role in modulating the immune environment than only through direct androgen receptor activation on immune cells.
The next step will be to dissect the underlying mechanisms of the immune alterations, and to assess if these do contribute to the reproductive complications and metabolic comorbidities of PCOS. This will include further characterization of immune cell changes to understand how their function could be affected and link these to reproductive and metabolic mechanisms.
The researchers also want to understand how the effect of androgens on eosinophils and mature NK cells is mediated, since neither of them expresses the androgen receptor.
More information: Sara Torstensson et al, Androgens Modulate the Immune Profile in a Mouse Model of Polycystic Ovary Syndrome, Advanced Science (2024). DOI: 10.1002/advs.202401772