This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Study uncovers how cancer stem cells spread and resist treatment
NDORMS researchers have identified a critical axis that controls the formation and behavior of cancer stem cells (CSCs), a subpopulation of cells that influence how lethal the cancer can be, its resistance to chemotherapy, and its ability to spread to other parts of the body.
The findings, published in Nature Communications, could pave the way for more effective cancer treatments.
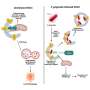
The research, led by Associate Professor and CRUK career development fellow Siim Pauklin from NDORMS, focused on understanding the intricate interplay between cell-autonomous (mechanisms inside cells that function independently of signals coming from outside the cells) and non-cell-autonomous (external signals from outside cells) pathways that govern the stem cell-like characteristics of cancer stem cells (CSCs) in pancreatic cancer.
CSCs have also been discovered in the brain, breast, colon, esophagus, liver, lung, ovarian, prostate, stomach and thyroid cancers, among others.
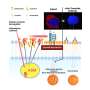
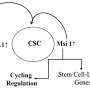
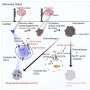
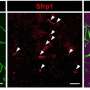
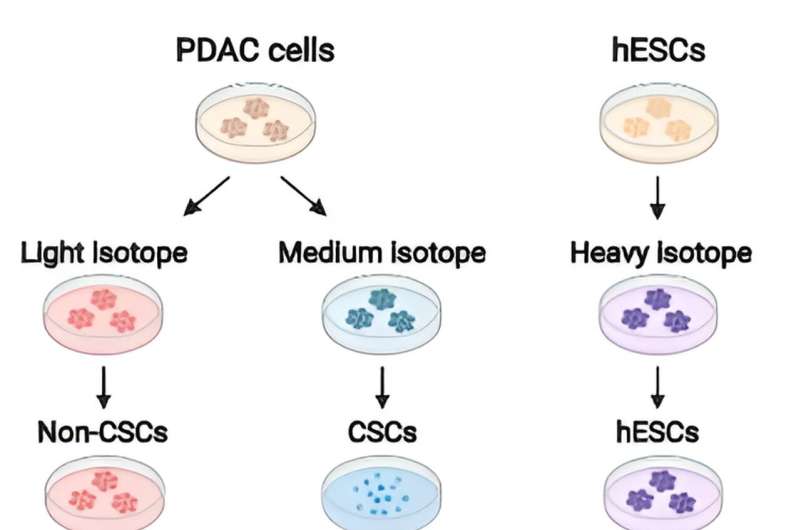
Using a quantitative proteomic approach to analyze the set of proteins in the extracellular space (the secretome) of pancreatic CSCs, the research reveals how a complex axis involving the retinoblastoma (RB) proteins and the E2F transcription factors controls the production of signaling molecules that promote CSC formation and chemoresistance.
"We were surprised to find that the well-known cell cycle regulators pRb and E2F, which were previously thought to only have cell-autonomous effects, actually have a non-cell-autonomous role in controlling the secretion of key WNT ligands that drive the stem cell-like properties of CSCs," said Siim. "This work provides important insights into the non-cell autonomous mechanisms that allow cancer stem cells to thrive and evade therapy."
The researchers discovered that the E2F1 and E2F4 transcription factors induce the expression of WNT ligands, such as WNT7A, WNT7B, WNT10A, and WNT4, while the pRb and RBL2 proteins act to reduce their expression. This delicate balance is disrupted in cancer cells harboring KRAS mutations, leading to the aberrant secretion of these WNT ligands and the activation of the WNT/β-catenin signaling pathway in CSCs.
"The interplay between cell-autonomous transcriptional regulators and non-cell-autonomous signaling pathways is a crucial, yet often overlooked, aspect of cancer stem cell biology," Siim said. "Our findings highlight the importance of targeting these extracellular communication networks to effectively eliminate this resilient population of cells."
The study's insights could pave the way for the development of more effective cancer therapies, as targeting the identified pathway could potentially disrupt the self-renewal, chemoresistance, and metastatic potential of CSCs, which are often responsible for treatment failure and disease recurrence.
More information: Chao-Hui Chang et al, The pRb/RBL2-E2F1/4-GCN5 axis regulates cancer stem cell formation and G0 phase entry/exit by paracrine mechanisms, Nature Communications (2024). DOI: 10.1038/s41467-024-47680-z