New MS drug proves effective where others have failed
A drug which 'reboots' a person's immune system has been shown to be an effective treatment for multiple sclerosis (MS) patients who have already failed to respond to the first drug with which they were treated (a 'first-line' therapy), as well as affected individuals who were previously untreated. The results of these two phase III clinical trials were published today in the journal The Lancet.
The new studies, sponsored by Genzyme (a Sanofi company) and Bayer Schering Pharma, showed that alemtuzumab significantly reduces the number of attacks (or relapses) experienced by people with MS compared to interferon beta-1a (known commercially as Rebif). This was seen both in patients who had not previously received any treatment (drug-naïve) and those who have continued to show disease activity whilst taking an existing treatment for MS.
In the CARE MSII trial, confined to patients who had recently relapsed on a licenced therapy, new episodes were reduced by 49 per cent more than that achieved by the current standard treatment for MS, interferon beta-1a. Over a two year period, 65 per cent of patients on alemtuzumab compared to 47 per cent of patients on interferon remained relapse free. Additionally, alemtuzumab reduced the risk of acquiring disability by 42 per cent compared to interferon: disability worsened in 20% of interferon patients and 13% of alemtuzumab patients. Moreover, at the end of the study, on average, patients taking alemtuzumab had less disability than when they started the trial whereas those on interferon had experienced worsening disability.
"Our research shows the transformative effect that alemtuzumab can have for people with MS. Patients who continue to show disease activity while on their initial therapy are especially difficult to treat. Now, we have shown that alemtuzumab works where first-line drugs have already failed. It not only reduces the chances of disability associated with MS but may even result in long-term clinical improvements," said Professor Alastair Compston from the University of Cambridge, principal investigator on both studies and Chair of the Steering Committee which oversaw these and earlier clinical trials.
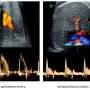
For the study (CARE MS II), researchers recruited 840 MS patients (of whom 628 were used for the principal analysis) who had been treated previously but had recently relapsed during their therapy. They were divided in a ratio of 2:1 to receive either alemtuzumab or interferon beta-1a. Their disability was then assessed every three months for two years by a researcher who was not made aware of which drug the patient had received. Additionally, they had annual scans to assess the lesions and brain shrinkage caused by MS.
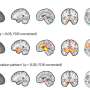
Alemtuzumab was found significantly to reduce the risk of having another relapse of MS or becoming disabled over the next two years, compared to one of the most effective licensed therapies for similar cases of MS, interferon beta-1a. Additionally, most patients on alemtuzumab experienced an improvement in disability, which was not seen after treatment with interferon-beta 1a. The brain scans showed that alemtuzumab not only reduced the number of new lesions, compared to interferon beta-1a, but also reduced the rate of brain shrinkage that occurs in MS as tissue is damaged.
"Although other MS drugs have emerged over the last year – which is certainly good news for patients – none has shown superior effects on disability when compared to interferon except alemtuzumab. Additionally, no other treatment has led to improvements in disability," said Dr Alasdair Coles, lead author of the paper and a clinician from the University of Cambridge.
MS is an autoimmune disease in which the body's immune system mistakenly attacks nerve fibres and their protective insulation, the myelin sheath, in the central nervous system. The resulting damage prevents the nerves from 'firing' properly and ultimately leads to the loss of the nerve fibre, resulting in physical and cognitive disabilities. The results of a second phase III clinical trial (CARE MS I) also examined the effectiveness of alemtuzumab against the drug interferon beta-1a but in 581 drug-naïve patients.
It found that alemtuzumab reduced the number of attacks experienced by people with relapsing-remitting MS by 55 per cent over and above that achieved by interferon beta-1a. Over a two year period, 78 per cent of patients on alemtuzumab compared to 59 per cent of patients on interferon remained relapse free. The proportion of patients experiencing worsening of disability on this trial was slightly lower after alemtuzumab than interferon beta-1a, but this result was not statistically significant.
For both studies, the principal side-effect of treatment with alemtuzumab was the development of other autoimmune diseases. During the trials, roughly 20 per cent of patients developed thyroid autoimmunity and 1 per cent developed an immune thrombocytopenia. Previous work has shown that up to 30 per cent of patients may develop autoimmune thyroid disease over time.
Laboratory work of the Cambridge research team, led by Dr Coles, is investigating how to detect people who are susceptible to these side-effects (funded by the Freemasons' Grand Charity and the Multiple Sclerosis Society in the UK). Additionally, they are currently recruiting for a trial of alemtuzumab in combination with a novel drug to reduce the risk of autoimmune diseases developing as a side-effect of taking alemtuzumab, funded by the Moulton Charitable Foundation and the Medical Research Council (MRC). Both Professor Alastair Compston and Dr Alasdair Coles are supported by the NIHR Biomedical Research Centre, Cambridge.
"Although alemtuzumab causes potentially serious side-effects, these can be identified and treated provided a monitoring schedule is carefully followed. Additionally, we think that we can identify which patients are at risk of autoimmune disease after alemtuzumab, and we are currently recruiting for a clinical trial which will explore whether we can use a drug to reduce the risk of autoimmunity in those at highest risk," said Dr Coles.
This brings to conclusion a unique programme for the development of a drug for MS that began in Cambridge in 1991. Never before has an MS drug been tested in clinical outcomes against such a high hurdle, an active first-line drug, in both one phase II trial and two phase III trials; and no drug for MS has been shown to be more effective, in both reducing the risk of disability and reducing the rate of brain atrophy, when compared to another active treatment.
Decisions on drug licences for alemtuzumab (known commercially as Lemtrada) by the European and US regulatory authorities are expected in 2013.
MS affects almost 100,000 people in the UK, 400,000 in the United States and several million worldwide.
More information: The papers 'Randomized Trial of Alemtuzumab in Patients with Multiple Sclerosis Relapsing on Disease-Modifying Therapy' and 'A Randomized, Rater-Blinded Study of Alemtuzumab Versus Interferon Beta As Initial Treatment For Relapsing-Remitting Multiple Sclerosis' will be published in the 1 November edition of The Lancet.