Neutrons used to study model vascular systems
In what may be the first use of neutron scattering to study complex bio-medical systems under dynamic conditions, Los Alamos researchers and collaborators mimicked blood flow by engineering a layer of human endothelial cells (the cells that cover the inner surface of blood vessels) and subjecting them to shear stress. Simultaneously, the team used neutrons at the Lujan Neutron Scattering Center's Surface Profile Analysis Reflectometer (SPEAR) to understand changes in the cell's properties. The technique, which relies on neutron reflectometry to reveal the behavior and composition of the cells, provides a new means to explore conditions that affect human vascular health. The American Journal of Physiology-Lung Cellular and Molecular Physiology has published the research.
Significance of the research
Because the vascular system is under constant fluid mechanical stress as a fluid conduction system, mechanical forces influence endothelial cell function and response. The endothelial cells are key in controlling the resistance of blood flow in arteries and veins, as well as blood coagulation. These cells are involved at nearly every level of vascular function as well as pathology. For example, endothelial cells and fluid mechanical shear stress are involved in atherosclerosis, a pathologic buildup of lipids in arterial walls that causes hundreds of thousands of deaths each year from heart attacks and strokes.
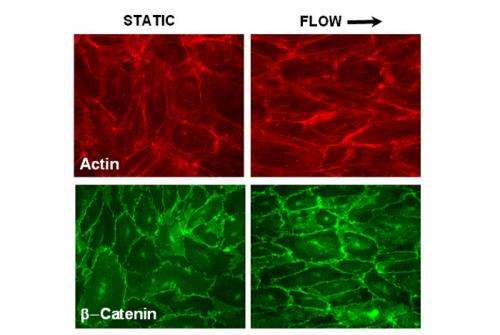
Most studies in endothelial mechano-biology have focused on single cells and even single molecules and protein complexes. However, the collective behavior of the monolayer of cells has been less well explored. It is clear that endothelial cells make strong lateral connections between each other and that these connections change as a function of mechanical stress.
In this work, the team examined the structure and composition of the boundary layer between the endothelial cells and the supporting substrate. The thickness of such a layer—its composition (protein rich vs. fluid rich) and how its composition and structure changes as a function of shear stress—has never been explored. By shining a beam of neutrons on the monolayer of cells and measuring the reflected radiation, the researchers revealed these long-awaited details.
The research measured living tissue adhesion strength under dynamic conditions. The study is the first to provide a direct measure of endothelial monolayer adhesion under physiologic shear stress conditions. The strength of neutron reflectometry is its non-perturbative nature and ability to probe large surface areas with nanometer resolution. Using endothelial monolayers, the team showed that physical models of soft-adhesion could describe large-scale tissue adhesion, which broadens their scope of application far beyond the single cell realm.
Research achievements
The team made neutron reflectometry measurements at 25 C and 37 C (physiologic temperature) of endothelial cells growing on quartz. At 25 C, most active biology is suppressed, and the endothelial monolayer responds as a soft matter physical system. However, at physiologic temperature, the full richness of cell biology is available to the system. Shear stress causes major redistribution of proteins involved in cell-cell and cell-substrate adhesion. Even on the relatively short biologic time scales (four hours) relevant to the time scale of the neutron experiments, the scientists observed cell-cell and focal adhesions on endothelial cells. This localization of adhesion proteins may promote linkage between the internal framework of the cell and extracellular matrix, forming enhanced adhesion zones. The researchers concluded that shear stress at 37 C causes localization of specific proteins, which may alter the adhesion potential in favor of overall increased adhesion.
More information: "Tuning endothelial monolayer adhesion: a neutron reflectivity study." Luka Pocivavsek, Ann Junghans, Noureddine Zebda, Konstantin Birukov, Jaroslaw Majewski. American Journal of Physiology - Lung Cellular and Molecular Physiology, Jan 2014,306(1)L1-L9; DOI: 10.1152/ajplung.00160.2013