Wound-healing role for microRNAs in colon offer new insight to inflammatory bowel diseases
A microRNA cluster believed to be important for suppressing colon cancer has been found to play a critical role in wound healing in the intestine, UT Southwestern cancer researchers have found.
The findings, first discovered in mice and later reproduced in human cells, could provide a fresh avenue for investigating chronic digestive diseases and for potentially repairing damage in these and other disease or injury settings.
"We identified a novel role for microRNAs in regulating wound healing in the intestine. This finding has important implications for diseases such as ulcerative colitis and Crohn's disease and may be relevant to wound healing mechanisms in other tissues," said Dr. Joshua Mendell, CPRIT Scholar in Cancer Research, Professor of Molecular Biology, and member of the UT Southwestern Harold C. Simmons Cancer Center.
Ulcerative colitis and Crohn's disease—the two most common inflammatory bowel diseases affecting an estimated 1.5 million in the U.S.—stem from an abnormal immune response, which results in the body mistakenly attacking cells in the intestine. The resulting chronic injury to the colon also is considered a risk factor for colon cancer. Understanding the cellular pathways involved could eventually lead to potential therapeutic treatments.
MicroRNAs serve as brakes that help regulate how much of a protein is made, which, in turn, determines how cells respond to various stimuli. Approximately 500 to 1000 microRNAs are encoded in the genomes of mammals. Dr. Mendell's laboratory studies how these tiny regulators work normally and how diseases such as cancer arise when they function in an abnormal manner.
These latest findings, which appear in the journal Cell, focus on two microRNAs: miR-143 and miR-145. While there is extensive literature implicating these microRNAs in colon cancer, little is known about their natural function in the colon. So Dr. Guanglu Shi, postdoctoral researcher in Molecular Biology, and other researchers began their five-year investigation by removing or "knocking out" the gene that produces these two microRNAs in mouse models.
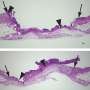
The researchers found that the cells that normally increase their growth to make repairs, called epithelial cells, fail under stress in the knockout animals. Epithelial cells line the intestines where food is digested, separating the contents from the rest of the body and absorbing needed nutrients.
"The epithelial cells of the colon normally proliferate quickly to fill in the wounds from an injury. Without these microRNAs, the epithelial cells are unable to switch into this repair mode, so they never heal the wounds and the mice are not able to survive," Dr. Shi said.
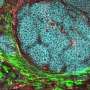
In addition, the research upended traditional thinking about where the tiny microRNAs reside, discovering to everyone's surprise that they reside in supporting cells, called mesenchymal cells, instead of the epithelial cells themselves as previously thought.
"This was surprising because colon cancers derive from the epithelial cells, so it was assumed that the microRNAs must function within them," Dr. Mendell said. "If these microRNAs do participate in colon cancer, they must do so by acting from outside the epithelium."
Identifying the accurate location of the microRNAs is essential to locating the pathways they regulate and eventually, to determining whether they can be manipulated for therapeutic purposes.
Dr. Mendell's team collaborated with a group of surgeons at UT Southwestern including Dr. Joselin Anandam, Assistant Professor of Surgery, Dr. Abier Abdelnaby, Assistant Professor of Surgery, Dr. Glen Balch, Assistant Professor of Surgery, and Dr. John Mansour, Assistant Professor of Surgery, and Dr. Adam Yopp, Assistant Professor of Surgery, who provided human tissue specimens. "The ability to work closely with an outstanding clinical team enabled us to confirm that our findings in mice extend to humans," Dr. Mendell said.
In addition, the researchers say they have worked out one of the many pathways regulated by these microRNAs, called the insulin-like growth factor signaling pathway.
"This pathway is involved in many different processes in the body, but one function is to stimulate wound healing responses," Dr. Mendell explained. "Increasing the amount of insulin-like growth factor signaling improves wound healing in the intestine."
Knocking out miR-143 and miR-145 counteracts that effect.