Small RNAs can prevent spread of breast cancer
Howard Hughes Medical Institute researchers have identified small pieces of ribonucleic acid (RNA) that suppress the spread of breast cancer to the lungs and bone. The new research shows that the most invasive and aggressive human breast cancer tumors are missing three critical microRNA molecules. When the researchers put those molecules back into human breast cancer tumors in mice, the tumors lost their ability to spread.
“The tiny RNAs prevent the spread of cancer by interfering with the expression of genes that give cancer cells the ability to proliferate and migrate,” said senior author Joan Massagué, a Howard Hughes Medical Institute researcher at Memorial Sloan-Kettering Cancer Center.
The researchers said their new research findings add to the growing number of cellular targets for drugs designed to prevent breast cancer metastasis. The scientists are also optimistic that their work could spur the development of new clinical tests to assess the likelihood that breast tumors will metastasize. Metastasis of breast cancer is the leading cause of death from the disease.
Metastasis occurs when cells from a primary tumor break off and invade another organ. It is the deadliest transformation that a cancer can undergo, and therefore researchers have been looking for specific genes that propel metastasis. If they can identify distinctive metastatic gene programs for different cancers, it may be possible to slow or halt metastases by targeting the proteins produced by those genes.
Massagué, his postdoctoral fellow Sohail Tavazoie, and colleagues at Sloan-Kettering, published their findings in the January 10, 2008, issue of the journal Nature.
In the experiments reported in Nature, the researchers explored the role that microRNAs play in controlling the expression of genes that trigger the spread of breast cancer. MicroRNAs, which are no more than 20-22 nucleotides in length, play an important role in development, cancer, stress responses and viral infections. Unlike the large messenger RNA (mRNA) molecules that code for cellular proteins, the microRNAs regulate gene activity by repressing or enhancing the translation of mRNAs.
Messenger RNA molecules are the genetic templates for proteins. In constructing proteins, the mRNA template is transcribed from DNA genes and transported to the ribosomes – the cell’s protein factories that are large complexes of protein and RNA. Given the importance of mRNA as an information-carrying molecule, the machinery that regulates mRNA levels and destroys faulty mRNA is critical in ensuring that errors in the genetic code are not passed on to proteins.
Massagué and his colleagues have focused on identifying the driving forces behind the spread of breast cancer, and have, in previous work, identified distinct genetic signatures associated with metastasis to the bone and the lungs. They decided to explore the role of microRNAs in breast cancer shortly after other researchers published data showing that certain tumors, including breast cancers, had decreased levels of microRNA expression. These observations suggested to Massagué and others that loss of the gene-suppressing molecules might well play a key role in the growth and spread of tumors.
To follow up on that hunch, Tavazoie and others in Massagué’s lab set their sights on identifying microRNAs that were suppressed only in aggressive, metastatic human breast cancers. They first analyzed cultured metastatic human breast cancer cells to create a genetic profile of the array of microRNAs produced by the cancerous cells. When they compared this microRNA profile to that from non-metastatic cancer cells, they found that a small subset of microRNAs was greatly reduced only in the metastatic cells.
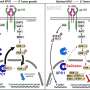
They next found that when they restored normal levels of three of these microRNAs—miR-335, miR-126 and miR-206—in the cultured human breast cancer cells, it greatly reduced the cells’ ability to spread to the lungs or bones of mice. They also obtained a sample of metastatic cells from a breast cancer patient and found that those cells had lost the same three microRNAs identified in the cell culture experiments.
To study a broader sample of cancers, the researchers measured levels of the microRNAs in cancer cells from 20 patients with breast cancer. They found lower levels of the microRNAs in patients whose breast cancers had metastasized than in those whose cancers had not metastasized.
Additional biological analyses revealed that miR-126 influences the proliferation rate of the metastatic cells, whereas miR-335 and miR-206 influence the cancer cells’ ability to migrate into lungs or bone.
The researchers found a particularly strong association between loss of miR-335 and cancer relapse, so they next sought to determine which genes were regulated by that particular microRNA. Their genetic analyses revealed a set of six genes whose activity greatly increased with loss of miR-335 in metastatic cells. Two genes in particular, SOX4 and TNC, were known to regulate cell migration, which is critical for cancer invasion of other tissues. When the scientists knocked down the activity of those two genes, they reduced the cancer cells’ ability to spread.
Taking the analysis a step further, Massagué’s team sifted through genetic data from 368 patients with breast cancer. Those data showed that tumors with a higher level of expression of the six miR-335-regulated genes were much more likely to metastasize than those tumors with a lower level of expression of the six genes.
According to Massagué, identification of the microRNAs and the metastasis-controlling genes they regulate could prove valuable for both breast cancer prognosis and treatment. “The gene signature we have identified could offer another tool for assessing the likelihood that a cancer will progress,” he said. “There are quite a few of these signatures, and the most useful ones are those that consist of genes that are not just markers, but are actually mediators of metastasis. And in this gene signature, we have identified just such a set of mediators.
“Secondly, these findings reveal still more genes that could be targets of drugs to interfere with metastasis,” he said. “Researchers in this field are currently accumulating an inventory of clinically significant genes to explore, and the set we identified has shown such significance.”
Massagué said his group is now designing experiments to ask whether the breast cancer genes regulated by the microRNAs they identified also control metastasis of other types of cancer. “We have basically opened a window into a major future inquiry into such genes,” he said. “And my hope is that this paper will lead to findings that are even more important and useful than the ones contained in this first installment.”
Source: Howard Hughes Medical Institute