Signaling to chromatin
(Medical Xpress) -- Scientists from the Friedrich Miescher Institute for Biomedical Research (FMI) in collaboration with their colleagues from the Department of Biosystems Science and Engineering of the ETH Zurich, describe how the signaling molecule JNK directly modifies histones to alter gene transcription. As JNK acts in a signaling pathway that is impaired in every third form of cancer, the results published in Nature Genetics open up a new pathway for kinase regulated gene expression and potential therapeutic intervention.
Signaling cascades take on the important task of communicating changes in the surroundings of the cell and initiating the appropriate responses. Cells perceive the changed environment through receptors on the surface that activate clearly defined signaling pathways. In the cell, these signaling cascades may diverge and activate a mesh of proteins usually through phosphorylation or de-phosphorylation. However, at the end of these cascades stands a transcription factor that adapts gene transcription to the changed circumstances. Critical for the functioning of the transcription factors is the accessibility of the DNA, which is controlled by epigenetic processes. Until recently these two domains have been clearly separated. There had been no evidence that signaling cascades also control the accessibility of the DNA. A recent publication from the laboratory of Dirk Schübeler, group leader at the Friedrich Miescher Institute for Biomedical Research and Professor at the University of Basel, proves this clear separation wrong.
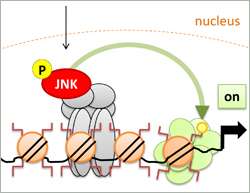
In a publication in the current issue of Nature Genetics Dirk Schübeler and his collaborators at the Department of Biosystems Science and Engineering (DBSSE) at the ETH in Basel, could show that one of the most abundant signaling molecules called JNK directly interferes with histones, the proteins that coil or de-coil the DNA.
In a genome wide analysis, they could show that JNK binds directly to a large number of promoters independent of the transcription factor AP-1, which is usually activated by JNK. The number of promoters that JNK binds to even increases as the cells differentiate. What is more, they could show that JNK directly phosphorylates Serine 10 at the tail of histone H3, a mark that is associated with relaxed chromatin and an active transcription state in non-dividing cells. Inhibition of JNK signaling reduced this histone phosphorylation and expression of JNK target genes.
"Signaling molecules interfering directly with chromatin are a very new concept," said Vijay Tiwari, first author of the publication and postdoc in Schübeler's lab. "It represents a new way how intracellular signaling molecules can influence gene expression and thereby cell fate."
The findings of the FMI scientists are also interesting in a more applied way. The signaling cascade in which JNK acts-the MAP kinase pathway-is one of the most important regulators of proliferation, differentiation and even cell death. It is often disrupted in diseases; almost a third of all cancers show defects in this pathway. "Our findings open up the field and expand the number of targets for a therapeutic intervention in these diseases," comments Tiwari, who will continue this research topic in his own laboratory that he will establish at the Institute of Molecular Biology (IMB) in Mainz, Germany.
More information: Tiwari VK, Stadler MB, Wirbelauer C, Paro R, Schübeler D, Beisel C, (2011) A chromatin-modifying function of JNK during stem cell differentiation, Nat Genet. 44(1):94-100. www.nature.com/ng/journal/v44/n1/full/ng.1036.html