Researchers show possible trigger for MS nerve damage
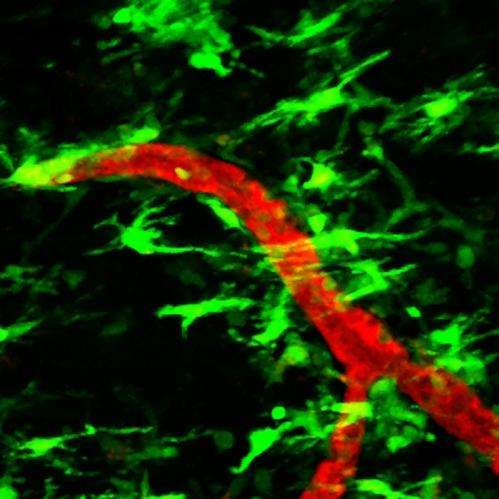
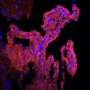
High-resolution real-time images show in mice how nerves may be damaged during the earliest stages of multiple sclerosis. The results suggest that the critical step happens when fibrinogen, a blood-clotting protein, leaks into the central nervous system and activates immune cells called microglia.
"We have shown that fibrinogen is the trigger," said Katerina Akassoglou, Ph.D., an associate investigator at the Gladstone Institute for Neurological Disease and professor of neurology at the University of California, San Francisco, and senior author of the paper published online in Nature Communications.
Multiple sclerosis, or MS, is thought to be an autoimmune disease in which cells that normally protect the body against infections attack nerve cells in the brain and spinal cord, often leading to problems with vision, muscle strength, balance and coordination, thinking and memory. Typically during MS, the immune cells destroy myelin, a protective sheath surrounding nerves, and eventually leading to nerve damage. The immune attack also causes leaks in the blood-brain barrier, which normally separates the brain from potentially harmful substances in the blood.
"Dr. Akassoglou has focused on the role of the blood-brain barrier leak in MS and has discovered that leakage of the blood clotting protein fibrinogen can trigger brain inflammation," said Ursula Utz, Ph.D., M.B.A., a program director at NIH's National Institute of Neurological Disorders and Stroke (NINDS).
Microglia are cells traditionally thought to control immunity in the nervous system. Previous studies suggested that leakage of fibrinogen activates microglia. In this study, Dr. Akassoglou and her colleagues used a cutting-edge imaging technique called two-photon laser scanning microscopy to watch what happens in an animal model of MS.
"Our results provide the first evidence linking leakage of fibrinogen to neuronal damage," said Dr. Akassoglou, "Vascular changes are the instigator of neurotoxicity."
Using a mouse model of MS, the researchers found that leakage of fibrinogen and microglial activation occurred days before nerve damage began, suggesting they occur in an early, pre-clinical stage of the disease.
During this period, microglia changed shapes and clustered around blood vessels along with other immune cells. Further experiments suggested that fibrinogen activated microglia by binding to a receptor, called CD11b/CD18, which caused the microglia to release reactive oxygen molecules that, in turn, damaged neurons. Inhibiting the binding of fibrinogen to the receptor prevented microglial activation and nerve damage.
Current treatments for MS are designed to suppress autoimmunity. The results from this study suggest that targeting the interaction between fibrinogen and microglia may be an effective alternative. In the mice, blocking fibrinogen's blood clotting activity prevented microglial activation and nerve damage.
"Current drugs target primarily downstream events. This interaction could be an upstream target that suppresses immunity and neurodegeneration," said Dr. Akassoglou.
In 2006, Dr. Akassoglou received a Presidential Early Career Award for Scientists and Engineers, which honors and supports the finest and most promising researchers early in their careers.
More information: Davalos et al., "Fibrinogen-induced perivascular microglial clustering is required for the development of axonal damage in neuroinflammation." Nature Communications, published online November 27, 2012. DOI: 10.1038/ncomms2230