March 25, 2013 report
Vesicle-attached ATP generator, not mitochondria, powers axonal transport
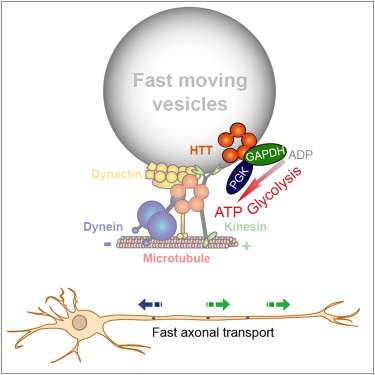
(Medical Xpress)—Neurons have developed elaborate mechanisms for transporting critical components, like transmitter-laden vesicles, down their axons to the synaptic terminations. An axon in a blue whale may be several meters long while those in M.mymaripenne, a wasp smaller than a paramecium, may be just microns long. Yet regardless of scale, these axons all seem to use similar molecular motors working on similar microtubule tracks to deliver vesicular cargo. In a paper recently published in Cell, researchers at INSERM, in France, have shown that the principle source of energy for these motors may not be the mitochondria as has been traditional assumed. Instead the ATP to power these motors appears to from vesicle-attached glycoloytic machinery, namely the enzyme GADPH (Glyceraldehyde 3-phosphate dehydrogenase).
In physics, the principle of conservation of energy (momentum as well) provides a way to solve many complex problems. Similarly in neurons, often the best way to try figure out how a particular mechanism works, is to analyze how the energy for it is sourced and dissipated. The INSERM researchers, who have studied the role of mitochondria in diseases like Huntington's disease in the past, noted that degeneration of axons is a principle pathology. In particular, they sought to find out whether the observed degeneration could be due to insufficient energy to power axonal transport. Curiously, they found that inhibiting the function of mitochondria, and hence the cell's main energy source, had no effect on the transport of vesicles.
As a preliminary study, the researchers used a sensor called Pervceval, previously developed by a group at Harvard, to assess the distribution of ATP in axons. Perceval is some sense, a special purpose nanomachine. It is constructed as a GFP fusion protein with built-in protein logic that enables it to precisely measure the ratio of ATP to ADP in cells. Using Perceval, the researchers found that this ratio was uniform in the axons of drosophila despite the observation that the distribution of mitochondria were not. Mitochondria, in myelinated axons at least, tend to congregate at the nodes of Ranvier, where energy demands would be expected to be high during spiking activity.
The researchers then went on to show that when GADPH, a ubiquitously expressed "housekeeping gene" was tampered with, axonal transport of vesicles was disrupted. GADPH is one of the enyzmes of the glycolytic pathway, and it generates a single ATP per reaction. The researchers also showed that transport could be restored by adding back a synaptotagmin-GADPH fusion protein. Synaptotagmin is normally is targeted to vesicles for transport down to synapses. These experiments suggested that the vesicles were using GADPH for transport, a conclusion which parallels other studies indicating that GADPH, rather than mitochondria, powers transmitter uptake into vesicles at nerve endings. The failure of previous efforts to improve mouse neurodegenerative models of ALS by increasing mitochondrial mobility may be in part explained by these studies.
GADPH is also known to interact with huntingtin, a protein that when mutated is a cause of Huntington's disease. The INSERM researchers were able to show that huntingtin is the likely link which joins GADPH to the vesicle membrane. Whether this is the main mechanism of the disease would need further work to demonstrate. Another unanswered question is whether GADPH bound to the microtubules would be a significant source of the ATP for vesicle transport.
It is interesting to note that the motors involved with transporting mitochondria in axons, perhaps not surprisingly, use mitochondrial ATP to do so. Mitochondria also use the same motor protein, kinesin-1 for transport. Mitochondrial often seem to have a mind of their own—their motions are much more elaborate than that of vesicles. Video of mitochondrial transport shows that they move more continuously up and down neurites, sometimes fusing, or even dividing, with the daughter mitochondria heading off in opposite directions.
ATP accounting in axons, as anywhere in a cell, is a complicated matter. The NA-K pumps which maintain or restore membrane potential in active neurons are estimated to be the largest ATP consumers in the axon. Transport of vesicles, and mitochondria, may be intimately linked to spiking activity. Tonically active neurons would have greater turnover and hence higher demand for transport products at the synapses. Correlating transport with the spiking activity of the neuron would give additional insight into how things really work in the axon.
When it comes to ATP, everyone in the cell seems to have their hand out. Frequently the efforts to quantitatively account for the energetic needs of each user in the cell come up short. The generation and flow of energy in cells involves many processes beyond ATP hydrolysis. For future studies, the authors would also like to investigate whether GADPH is required for non-vesicular transport, to try to understand how, for example, RNA granules end up in the synapse. Studies like this one, which give greater insight into how ATP is generated and used, shed important light on both healthy and disease states of the cell.
More information: 1. Vesicular Glycolysis Provides On-Board Energy for Fast Axonal Transport, Cell, Volume 152, Issue 3, 479-491, 31 January 2013. DOI: 10.1016/j.cell.2012.12.029
2. Cell biology: Alternative energy for neuronal motors, Nature 495, 178–180, 14 March 2013. doi:10.1038/495178a
© 2013 Medical Xpress