Breakthrough discovery into the regulation of a key cancer drug target
There's not much difference between what makes a man and what makes his beer - at least at the molecular level - according to a new study led by Professor John Schwabe at the University of Leicester.
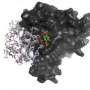
Scientists used a powerful technique called protein crystallography to look at 3D structures of protein complexes purified from cultured human cells. They discovered that a family of complexes, that switch off gene expression, is regulated by small signalling molecules called inositol phosphates.
This latest study shows that this mode of regulation is conserved from yeast right through to man and is a fundamental process for life.
This discovery is potentially important for developing anti-cancer drugs since these complexes are emerging as effective cancer treatment targets.
The research was funded by the Wellcome Trust and involved collaboration with scientists from the University of Oxford. It was published online ahead of print (July 11) in the journal Molecular Cell.
The importance of this work is emphasised by the award, in November 2012, of £2.4 million in the form of a prestigious "Wellcome Trust Senior Investigator Award" to Professor John Schwabe to continue and develop this area of research.
Professor Schwabe said: "Gene regulation is an essential biological process that occurs in every cell in the body to control which proteins are expressed in particular tissues. We have been studying enzymes called Histone Deacetylases (HDACs) that regulate this process. The action of HDACs causes the DNA to become more tightly packaged and thus switches off genes by making them unavailable for expression. HDACs are recruited to DNA by a number of accessory proteins (corepressors) that target the enzyme to the correct gene."
In 2012, the research team announced in Nature that they had discovered a completely new and unexpected mechanism through which one particular HDAC complex (HDAC3 bound to its companion protein SMRT) is regulated.
Professor Schwabe said: "We wanted to ask whether this mode of regulation might apply to other HDAC complexes and be a general principle for HDAC regulation. Now we have taken this investigation several steps forward.
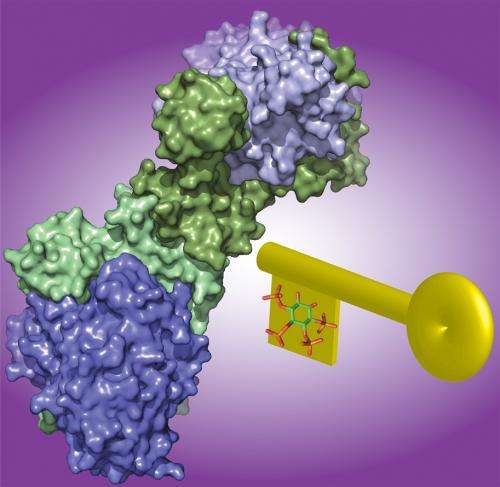
"We have solved the first structure of HDAC1 bound to one of its corepressor proteins (MTA1 – Metastasis-associated protein 1). This new structure allows us to examine how the proteins interact at the atomic level and reveals molecular details that have allowed us to probe the interaction surface.
"We have identified that there is an inositol phosphate-binding site at the interface between the two proteins, and through functional studies, we have confirmed that inositol phosphates are key regulators throughout this class of enzyme.
"This establishes that inositol phosphate regulation is a general paradigm that is conserved from yeast to man. In addition, we have shown that two MTA1 molecules come together to recruit two HDAC enzymes, and we suggest that MTA1 is critical for recruiting HDAC1 to DNA."
Professor Schwabe also emphasised that his research team is very privileged to have secured further funding from the Wellcome Trust to support this programme of research for the next 7 years: "We expect to gain an in depth insight into the role of inositol phosphates in the regulation of class I histone deacetylases and the specificity conferred to histone deacetylases by their recruitment into large multi protein complexes".
The research team used a cutting edge technique (protein crystallography) to look at the 3D structure of proteins involved in gene regulation. They optimised the conditions needed to grow microcrystals and took these to the UK's powerful synchrotron (Diamond Light Source, Oxford) to obtain diffraction data. These data are used to build images of the proteins, and for the first time, the team has been able analyse how the proteins interact with each other.
"With this new level of molecular detail we are exploring the design of novel HDAC inhibitors to target of the HDAC-corepressor interface. These may provide the foundation for future drug development and may lead to new anti-cancer therapies".