Study finds that microbes influence B-cell development in the gut
Gut bacteria exert a dramatic, systemic effect on the development of the immune system's B-lymphocytes, according to a new mouse study by researchers at Boston Children's Hospital. While influences of gut bacteria on T-lymphocytes have been noted before, this is the first time that researchers have documented early B-cell development in the gut and that microbes influence this process.
The study team—led by Duane Wesemann, MD, PhD, and Frederick Alt, PhD, of Boston Children's Hospital's Program in Cellular and Molecular Medicine (PCMM)—reported their findings online Aug. 21 in Nature.
Starting at birth, the immune system programs immature B-cells to produce antibodies against a wide array of potentially pathogenic antigens by shuffling genes for different antibody components. This shuffling process, called V(D)J recombination, depends on a factor called RAG, which results in an immense number of B-cells that collectively are able to respond to a diverse repertoire of antigens that the immune system has yet to encounter.
V(D)J recombination programs each individual B-cell to produce a single kind of antibody that will work against a single antigen. Because this shuffling process is random, some newly generated B-cells make self-reactive receptors. If this happens, the cells continue the RAG-mediated shuffling, a process called editing, which replaces a self-reactive receptor with one that is not self-reactive.
Several recent studies focused on T-cells have suggested that gut microbes have a range of effects on the immune system.
"We were looking at B-cells in the gut because we had previous data suggesting that certain B-cell lymphomas arise from cells that undergo editing in gut-associated lymphoid tissues," says Alt, director of the Boston Children's PCMM, and the Charles A. Janeway Professor of Pediatrics at Harvard Medical School. "However, we didn't expect to also find an active process of early B-cell development and antibody diversification going on in the gut."
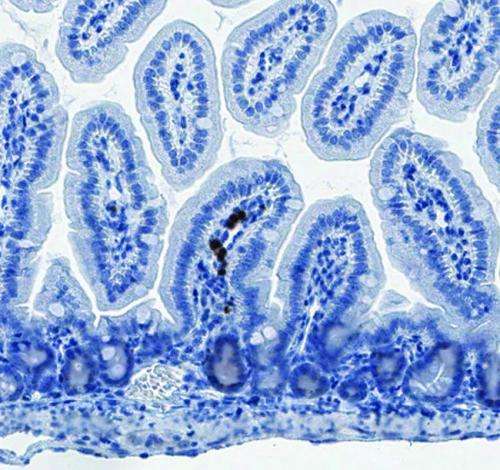
The team found that immature B-cells within the lamina propria portion of the gut were actively shuffling antibody genes, as measured by RAG levels and V(D)J recombination intermediates. In fact, the level of gene shuffling within gut B-cells was similar to that in B-cells developing in the bone marrow, suggesting there also was primary B-cell development in the gut. These results stand counter to current dogma on B-cell development in mice and humans.
"Sheep, rabbits and chickens develop their primary B-cell repertoire in the gut," says Wesemann. "Previously, primary antibody diversification had only been demonstrated in the bone marrow of humans and mice; so the finding that the process also occurs in the mouse gut was surprising."
"It could be that the gut serves as another location for primary B-cell development in the mouse in addition to the bone marrow," Alt adds. "Others have found immature B-cells in the lamina propria in humans, which based on our mouse studies suggests that gut B-cell development also might occur in humans."
When the team looked more closely, they saw that the diversity of the B-cell repertoire in the gut differed markedly from that in the bone marrow, as measured by sequencing- shuffled antibody genes in B-cells from both locations. The level of shuffling was the same, but the nature of the actual rearrangements differed dramatically between the two sites. The study also showed that at least some of the repertoire differences may have been generated by editing in gut B-cells.
A striking aspect of the discovery was that the amount of gene shuffling occurring in immature B-cells from weaning-age young mice changed dramatically depending on whether the mice were colonized with normal intestinal bacteria or were raised in a germ-free environment. Immature B-cells from mice with colonized intestines exhibited significantly more developing B-cells than those from germ-free animals, findings that held true in the gut, bone marrow and spleen.
Overall, the study suggests that gut microbes not only regulate T-cell activities but also influence those of B-cells.
More information: dx.doi.org/10.1038/nature12496