To beat leukemia, boost cell signaling, study suggests
A new study of acute lymphoblastic leukemia (ALL) led by UC San Francisco researchers puts an intriguing new twist on anti-cancer strategies. Rather than inhibiting cellular signals that arise from a cancer-driving gene—the most common mode of action of modern targeted therapies—the researchers markedly increased signaling in cancerous cells, causing the cells to self-destruct and eliminating ALL in mouse models of the disease.
Senior author Markus Müschen, MD, PhD, professor of laboratory medicine and co-director of UCSF's Hematologic Malignancies Program, said the success of this unconventional approach, reported today in the March 23, 2015 online issue of Nature, is due to particular characteristics of B cells, the bone-marrow-derived immune-system cells that are affected in ALL.
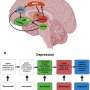
When fully mature, B cells carry receptors that detect potential bacterial or viral invaders and trigger the deployment of defensive antibodies. But as they develop, B cells are subject to what might be called a Goldilocks principle: If signals from the B cell receptor in a given cell are too weak to be effective, that cell is eliminated from the body; if signaling is too strong, which raises the danger of an immune reaction to the body's own tissues, the cell is likewise eliminated. Only B cells that are "just right" in terms of signaling strength become full-fledged players in the immune system.
"All cancer cells have receptors that generate signals, but B cells are special because they have to fit through a very narrow window of signaling strength. Weak signals won't sustain proliferation and survival, and the cells will die, which is also the case in breast or lung cancer cells driven by oncogenes," said Müschen. "But the upper threshold of B cells is unique: if the signal is too strong, there is a checkpoint that will mark those cells for elimination as a measure of protection against autoimmunity."
Usually emerging in children between 2 and 5 years of age, but also occurring in adults, ALL occurs when B cells proliferate out of control. In children many cases of ALL are successfully treated with high doses of chemotherapy, but in about a quarter of adult patients with ALL the presence of a genetic abnormality known as the Philadelphia chromosome (abbreviated Ph+) can render the disease resistant to conventional treatment.
In Ph+ ALL, a cancer-causing enzyme called a tyrosine kinase mimics active B cell receptor signaling, and does so at just the right level to allow for steady and continuous growth of the leukemia cells, which allows them to proliferate. About 15 years ago, the Food and Drug Administration approved the targeted drug imatinib (trade name Gleevec), which tamps down the activity of the tyrosine kinase and lowers B cell signaling to a level where Ph+ B cells are weeded out.
Though imatinib has been a significant advance in the treatment of ALL, patients develop resistance to that drug as well, and to overcome this problem pharmaceutical companies are actively developing more potent tyrosine kinase inhibitors.
In the new study, however, first authors and UCSF postdoctoral fellows Zhengshan Chen, PhD, and Seyedmehdi Shojaee, PhD, tried the opposite approach. Working with an international team of researchers, Chen and Shojaee found that if they used a novel drug that unleashed B cell receptor signaling, they could increase signaling to the point that it exceeded the checkpoint that governs the body's anti-autoimmunity mechanisms.
In experiments using this compound in mice, imatinib-resistant human Ph+ ALL cells were eliminated through a process of rapid and massive cell death, which significantly prolonged the survival of the mice. This effect was highly selective: only Ph+ cells were marked for destruction, suggesting that a low-toxicity B cell receptor-targeted drug compound could be developed to overcome imatinib resistance without affecting normal B cells.
A short exposure to the drug was sufficient to commit the leukemia cells to cell death and to clear most of the disease burden from mice. Müschen said that the drug's fast action was encouraging, because it remains unknown whether prolonged B cell receptor hyperactivation is safe, which is why he and colleagues are now focusing on formulating hyperactivating drugs that could be administered for only a few hours.
"These experiments show that we can engage signaling checkpoints in a very short period of time, and that once these checkpoints are engaged the cell is irreversibly slated for death—it's a point of no return," Müschen said. "The next step is to work with medicinal chemists to make better ALL drugs that will overstimulate the B cell receptor pathway, and that could be used on a treatment schedule to elicit a very strong, but time-limited, spike in signaling to engage negative B cell selection."
More information: "Signalling thresholds and negative B-cell selection in acute lymphoblastic leukaemia." Nature (2015) DOI: 10.1038/nature14231