August 20, 2015 report
Horizontal transfer of mitochondria in sickness and in health
(Medical Xpress)—Two of the most enticing ideas in cells biology have recently converged to create a paradigm shift of epic proportions. The first is that not only is it possible for mitochondria to emigrate from their host cell, they are in fact exchanged among cells much more regularly than has ever been imagined. The second is that while happenstance mutations are clearly associated with different aspects of a litany of cancers, the canonical force consistently driving tumor initiation, progression, and metastasis is now broadly understood to be the metabolic fickleness of their mitochondria.
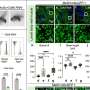
Mike Berridge is one of a handful of researchers firmly planted at the intersection of these two now ineluctable conclusions. As an author on a recent review in Cancer Research on the horizontal transfer of mitochondrial DNA (mtDNA), he adds much needed flesh to the first order simplification that cancer is merely a mitochondrial respiratory insufficiency. Most poignantly, in noting that the hidden force driving tumor-formation forward can more generally be understood to be the reacquisition of once lost mitochondrial function, new therapeutic opportunities immediately present themselves.
Of particular note Berridge found that the apparent need and ability of mitochondria-free primary tumor lines to re-assemble functional respirasomes, the supercomplexes responsible for respiration, differed according to cancer type. For example, breast cancer cells were found to have a unique 'threshold' level of respiration that was different from melanoma cells. Nover anticancer agents could in theory be designed to target specific components in more respiration-dependant cancer cells while leaving other cell types unscathed.
This kind of strategy may be slightly at odds with several current efforts we might only describe as a blanket restoration of respiratory function. This general picture, with many intrinsic unknowns still holding forth, is complicated by the finding that cancer cells acquiring mitochondria show chemoresistance. While the data shows that some threshold level of respiration is a prerequisite for tumor initiation, there must be functional aspects of mitochondrial acquisition at play beyond simple recovery of respiration.
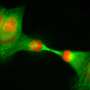
Some of the early studies of mitochondrial transfer only looked for the acquisition of mtDNA—the actual transfer of live, or at least whole, mitochondria was only inferred by the lack of any other suitable explanation for the mtDNA presenting itself in a strange cell. Typically these studies looked for the presence of unique versions of mitochondrially-encoded cytochrome b subunits in a background of cells which were originally homoplastic as far as their mitochondria. These days there is much more direct evidence of mitochondrial transfer in everything from bone and lung cells, to optic nerve axons. Berridge is even looking for evidence of transexudation from local astrocytes to synapses for the cases where transport down extremely long axons would seemingly be prohibitively slow to promptly reapportion fresh mitochondria to far-flung places of need.
An even clearer picture of transfer may be soon be had with new methodological advances. For example, nuclear-encoded green and red fluorescent proteins possessing mitochondrial import sequences should soon be available to researchers. Time lapse studies would then be able to distinguish whether whole cells are transiently fusing to bidirectionally swap organelles, or rather if there is only a more directed donation via endosomal fusion and uptake.
As far as general mitochondrial awesomeness, there are a few new studies (if at first seemingly unrelated), that hint at what might be next in all things mitochondria. Two recent papers in Cell have redefined the essential role of the mitochondrial electron transport chain in proliferating cells to simply be the synthesis of aspartate. In other words, these cells don't need the energy so much as they need the raw material for expanding their ranks, tumerigenic or otherwise.
If you had suggested a decade ago that parkinsonian flies could be cured by increasing the rate of fusion to change the morphology of their mitochondria just by spiking their diet with stearic acid people might look at you like you had two heads. Today, that particular science is par for the course. Control of mitochondrial morphology may be just one aspect to those supernumerary intangibles we alluded to above in noting that there may be more to the picture than just the standard respiratory biochemistry in controlling tumor development.
More information: Mitochondrial DNA in Tumor Initiation, Progression, and Metastasis: Role of Horizontal mtDNA Transfer, Cancer Research, Published OnlineFirst July 29, 2015; DOI: 10.1158/0008-5472.CAN-15-0859
Abstract
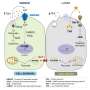
Mitochondrial DNA (mtDNA), encoding 13 out of more than 1,000 proteins of the mitochondrial proteome, is of paramount importance for the bioenergetic machinery of oxidative phosphorylation that is required for tumor initiation, propagation, and metastasis. In stark contrast to the widely held view that mitochondria and mtDNA are retained and propagated within somatic cells of higher organisms, recent in vitro and in vivo evidence demonstrates that mitochondria move between mammalian cells. This is particularly evident in cancer where defective mitochondrial respiration can be restored and tumor-forming ability regained by mitochondrial acquisition. This paradigm shift in cancer cell biology and mitochondrial genetics, concerning mitochondrial movement between cells to meet bioenergetic needs, not only adds another layer of plasticity to the armory of cancer cells to correct damaged mitochondria, but also points to potentially new therapeutic approaches.
© 2015 Medical Xpress