Trapping cells that tumors release into the bloodstream allows fast and sensitive genetic analysis

A simple and non-invasive method to detect cancer-related mutations without direct biopsy sampling of tumors has been developed by A*STAR researchers.
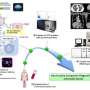
The technique uses a tailor-made filter to collect cancer cells circulating in the bloodstream after being shed from the tumor mass. These circulating cells can serve as a liquid biopsy. The study was led by Min-Han Tan and Jackie Y. Ying from the Institute of Bioengineering and Nanotechnology (IBN) of A*STAR, with collaborators from the Genome Institute of Singapore of A*STAR and other research centers in Singapore.
Genetic analysis of cancer cells is crucial for characterizing a cancer type and selecting the most suitable treatment options for a patient. Genetic results also play a role in monitoring the effectiveness of treatment and establishing a likely prognosis, explains Tan.
Many tumors shed cells into the bloodstream and several methods have been developed to try to capture these cells to assist in diagnosis, analysis and treatment. All previous attempts, however, have significant deficiencies, particularly in efficiently separating the cancer cells from other cells circulating in the blood.
"We used special microfabrication technology, which results in very uniform and precisely controlled pore size and depth for isolating the circulating cancer cells," explains Ying.
The silicon micro-sieves (see image) developed using this technique comprise a two-layered structure, with an upper thin porous membrane for cell filtration and a lower layer of thick honeycomb rings to support the membrane.
To prove the validity of their novel approach, the research team used blood samples taken from patients with bowel cancer, the most common type of cancer in Singapore. But, Tan says, their method is also likely to be applicable to a wide range of tumors, such as those in breasts, lungs and kidneys.
Having isolated the cancer cells, detailed genetic analysis was able to identify two specific types of mutation associated with bowel cancer. The analysis process begins directly on the sieve, and is completed within the sampling system, which significantly simplifies and speeds up the procedure. The new method also proved more sensitive than earlier alternatives, confirming that it could be of considerable clinical significance for improved diagnosis and treatment.
The team is now working towards developing the technology for clinical use.
IBN's non-invasive genetic test was licensed to AITbiotech Pte Ltd, a Singapore-based biotechnology company, in January 2015 for development into a ready-to-use test kit. AITbiotech provides molecular diagnostics and genomic services to hospitals and research laboratories.
More information: "Non-invasive sensitive detection of KRAS and BRAF mutation in circulating tumor cells of colorectal cancer patients." Molecular Oncology 9, 850–860 (2015). dx.doi.org/10.1016/j.molonc.2014.12.011