A deeper take on our sexual nature
The organs in our body may have a sexual identity of their own, new research suggests. The idea that our organs could be "male" or "female" raises the possibility that women and men may need different treatments as a result. The findings could also shed light on why it is that some cancers are more common in women, and others in men.
The study, published today in Nature, was carried out in fruit files by a team at the MRC Clinical Sciences Centre (CSC), based at Imperial College London.
"We wanted to ask a very basic question: whether it is just the cells of the sex organs of a fully developed organism that 'know' their sexual identity, or whether this is true of cells in other organs too - and whether that matters," said Irene Miguel-Aliaga, who led the research and heads the Gut Signalling and Metabolism group at the CSC.
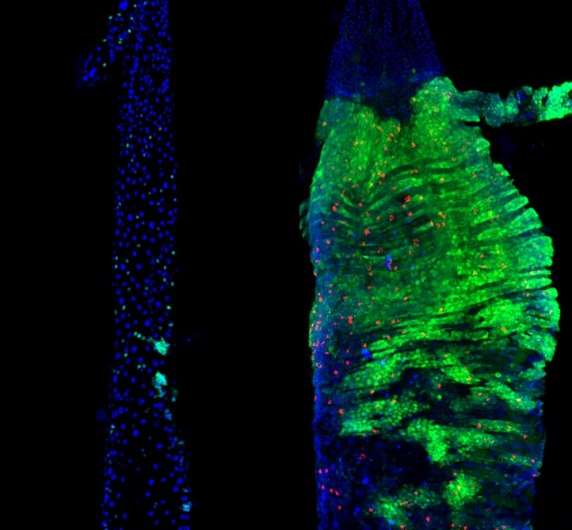
To do this, the CSC team examined stem cells in flies' intestines. They used genetic tools that allow them to turn genes "on" and "off" specifically in these cells. This allowed them to tailor the cells to be more "female" or more "male". When the team feminised or masculinised the flies' gut stem cells this changed the extent to which the cells multiplied. Female, or feminised cells were better able to proliferate.
This enhanced ability appears to allow the intestine in female flies to grow during reproduction. Miguel-Aliaga has previously shown that after mating, the female fly gut is re-sized and metabolically remodelled to sustain reproduction. She speculated at the time that if such enlargement also helps to ensure optimum nutrition for a developing fetus in humans, this may help to explain why women don't need to "eat for two" during pregnancy.
In the current study, the team found that the effect of feminising adult gut stem cells was reversible. "If we take a female fly and then in the adult we masculinise the stem cells in the intestine and wait, within three weeks the gut shrinks to the smaller, male-like size," said CSC and EMBO-postdoc, Bruno Hudry, who is first author on today's paper.
The team also found that the female intestine was more prone to tumours. "We find it's a lot easier to create genetically-induced tumours in females than in males. So we suspect there is a trade-off going on. Females need this increased plasticity to cope with reproduction, but in certain circumstances that can be deleterious and make the female gut more prone to tumours."
It was known that the gonads, or sex organs, of vertebrates retain considerable plasticity: adult ovary and adult testis cells in mice can trans-differentiate into their counterparts following just a single genetic change. So cells in the gonads must have their sexual identity continuously reinforced throughout their postnatal life.
The teams believes this to be the first time, however, that such plasticity has been demonstrated in adult cells outside the gonads. "If we now take the fly in which we've masculinised the stem cells, and mate this fly, we see that its gut is not re-sized in response to reproduction. So we think that what the stem cell sexual identity is doing is to confer differential plasticity on the female gut," Hudry said.
It may be obvious that males and females develop sex-specific organs as a fetus develops, but according to Miguel-Aliaga there has been an assumption that organs that are the same in both sexes function differently only because of different circulating hormones – for example, the oestrogen in women and testosterone in men that kick-in at puberty.
What's surprising is that in an adult organ found in both sexes, such as the intestine, differences remain, and that these are not due to either developmental history or circulating hormones. The benefit of using a fly model is that the team can masculinise or feminise individual adult stem cells and explore the outcome without changing the fly's developmental history or its circulating hormones.
In the course of this research, the team identified a potentially important new mechanism behind this sex switching, which they suggest may be operating in more of the organs and tissues of our bodies than has previously been recognised. Miguel-Aliaga sees this as an important finding of today's paper.
The formation of female or male characteristics involves a cascade of genetic events. "We saw that the sex determinants at the top of this cascade were active, but parts of the cascade that had previously been shown to be active, for example in the sex organs or during development, did not function in these stem cells. This told us a new branch of the sex determination pathway is at play."
Until now, it has been assumed that the only cells with a sexual identity are those in which this recognised cascade is active. "We have found a new mechanism that is independent of this, which potentially means that every cell in the fly has a sexual identity."
According to Miguel-Aliaga this raises the intriguing possibility that cells in many more fly organs than previously assumed may have their own sexual identity, and that this might be the case in people too.
"We want to know what this new branch is all about. We have found three genes that are important in the gut stem cells, and are intrigued to know whether these three genes play a similar role in cells outside of the gut in other body tissues as well." The team is keen to examine these three genes further, not least because people carry genes that are analogous to the three they've now highlighted as important.
Dr Des Walsh, head of the population and systems medicine board at the MRC, said: "This study is an interesting piece of biological research that extends our understanding of why male and female physiology is different, beyond the obvious.
"Further research is now needed to see how this finding translates to humans. If this intrinsic knowledge held by stem cells is indeed driving the way our organs behave, it could also influence the way these same organs respond to treatment."
More information: Bruno Hudry et al. The sexual identity of adult intestinal stem cells controls organ size and plasticity, Nature (2016). DOI: 10.1038/nature16953