Newly identified pathway links fetal brain development to adult social behavior
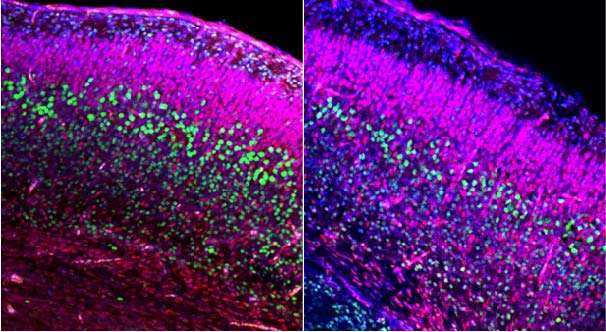
Fetal development has been known to play an important role in social interaction, a fundamental behavior found in nearly all organisms, and later adult social behaviors. Autism, a highly heritable neurodevelopment disorder that causes difficulties with social interactions, has been postulated to be caused by neuron overgrowth in the prenatal period, although the precise timing and cause of this overgrowth has been unknown. Researchers at Case Western Reserve University School of Medicine, UCSF School of Medicine and other institutions have recently uncovered abnormalities in embryonic brain development in mice, including transient embryonic brain enlargement during neuron formation, that are responsible for abnormal adult brain structures and behavioral abnormalities. These findings demonstrate a fetal origin for social and repetitive behavior deficits, as seen in disorders such as autism.
Using engineered mice, the researchers identified a critical period during embryonic brain development for the establishment of normal social behavior and were able to link this critical period with abnormalities in specific adult brain structures. After identifying these abnormalities, the researchers were then able to intervene and treat mice during fetal development. The embryonically treated mice progressed without adult behavioral deficits. Further studies will be required to determine the consequences of abnormal development of cortical neurons on adult brain circuitry and function, as well as possible therapeutic interventions. The results of the study were published in the current issue of Molecular Psychiatry.
"It is likely that abnormal brain development can cause long term alterations in brain circuitry that may later manifest as behavioral affective disorders in adults. By defining the pathway that connects embryonic development to adult social conditions, we were able to target the pathway in the embryo and provide a potential approach to stop the later development of abnormal behaviors. In the mice we treated, we were able to reverse the embryonic deficits that appear to lead to social and repetitive behavior disorders," said Anthony Wynshaw-Boris, MD, PhD, Chair, Department of Genetics and Genome Sciences, Case Western Reserve University School of Medicine and University Hospitals Case Medical Center. The work was initiated when he was a professor in the Department of Pediatrics at UCSF School of Medicine.
"We examined brains from the mouse embryos that developed social and repetitive behavioral abnormalities as adults since a number of children that develop autism display larger brains sooner after birth than typically developing children," said Haim Belinson, PhD, a postdoctoral fellow in the Wynshaw-Boris laboratory and first author of the Molecular Psychiatry paper. "We believe these findings may be relevant to the development of autism, and we are currently investigating this further."