December 1, 2016 report
Noninvasive microscopy technique allows for spotting skin cancer in mitochondria
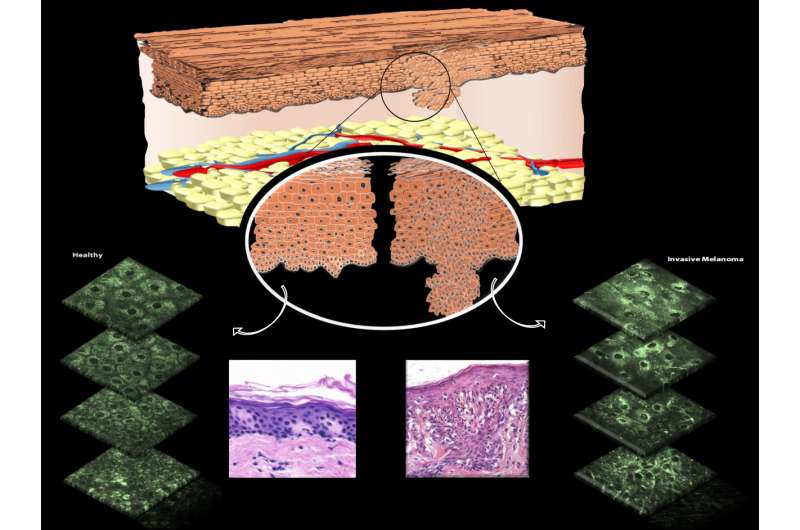
(Medical Xpress)—A team of researchers from several institutions in the U.S. and one in Spain has developed a new way to test for skin cancer in patients—one that does not rely on taking a biopsy. In their paper published in the journal Science Translational Medicine, the researchers outline their technique and describe how it is able to see irregularities in mitochondria.
Prior research has suggested that the development of tumors involves in part a disruption of the mitochondria network inside of cells. Prior research had also found that there is an enzyme inside of mitochondria called NADH that fluoresces when exposed to a certain wavelength of light. Noting that a type of high-powered multiphoton, laser-based microscope had recently been developed, the researchers sought to learn whether it could be used to detect skin cancer by noting mitochondrial disruptions—oddly arranged NADH could be spotted by shining a special light on them in skin cells in vivo. To facilitate the process, the team came up with an automated method of assessing whether mitochondria were organized in an abnormal way, layer by layer of skin.
After putting together all the components, the researchers enlisted the assistance of 10 patients with known skin cancer and four that were known to be cancer free. They then tested multiple skin sites on each of the volunteers. They report that the new method allowed them to see the cancer in those that had it and the absence of it in those that did not—all without having to make any cuts to the skin. To ensure the accuracy of their results, all of the volunteers were also subjected to traditional biopsies in which samples were sent to a lab for review.
More work will have to be done by the researchers and others in the field to prove the true accuracy of the technique, of course, but if it is as accurate as the initial results indicate, patients may soon be able to have their skin tested for cancer right in the doctor's office, doing away with the pain of a biopsy and the anxious wait on results from the lab.
More information: D. Pouli et al. Imaging mitochondrial dynamics in human skin reveals depth-dependent hypoxia and malignant potential for diagnosis, Science Translational Medicine (2016). DOI: 10.1126/scitranslmed.aag2202
Abstract
Active changes in mitochondrial structure and organization facilitate cellular homeostasis. Because aberrant mitochondrial dynamics are implicated in a variety of human diseases, their assessment is potentially useful for diagnosis, therapy, and disease monitoring. Because current techniques for evaluating mitochondrial morphology are invasive or necessitate mitochondria-specific dyes, their clinical translation is limited. We report that mitochondrial dynamics can be monitored in vivo, within intact human skin by relying entirely on endogenous two-photon–excited fluorescence from the reduced metabolic coenzyme nicotinamide adenine dinucleotide (NADH). We established the sensitivity of this approach with in vivo, fast temporal studies of arterial occlusion-reperfusion, which revealed acute changes in the mitochondrial metabolism and dynamics of the lower human epidermal layers. In vitro hypoxic-reperfusion studies validated that the in vivo outcomes were a result of NADH fluorescence changes. To demonstrate the diagnostic potential of this approach, we evaluated healthy and cancerous human skin epithelia. Healthy tissues displayed consistent, depth-dependent morphological and mitochondrial organization patterns that varied with histological stratification and intraepithelial mitochondrial protein expression. In contrast, these consistent patterns were absent in cancerous skin lesions. We exploited these differences to successfully differentiate healthy from cancerous tissues using a predictive classification approach. Collectively, these results demonstrate that our label-free, automated, near real-time assessments of mitochondrial organization—relying solely on endogenous contrast—could be useful for accurate, noninvasive in vivo diagnosis.
© 2016 Medical Xpress

















