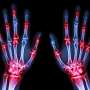
Baricitinib associated with significant improvement in RA
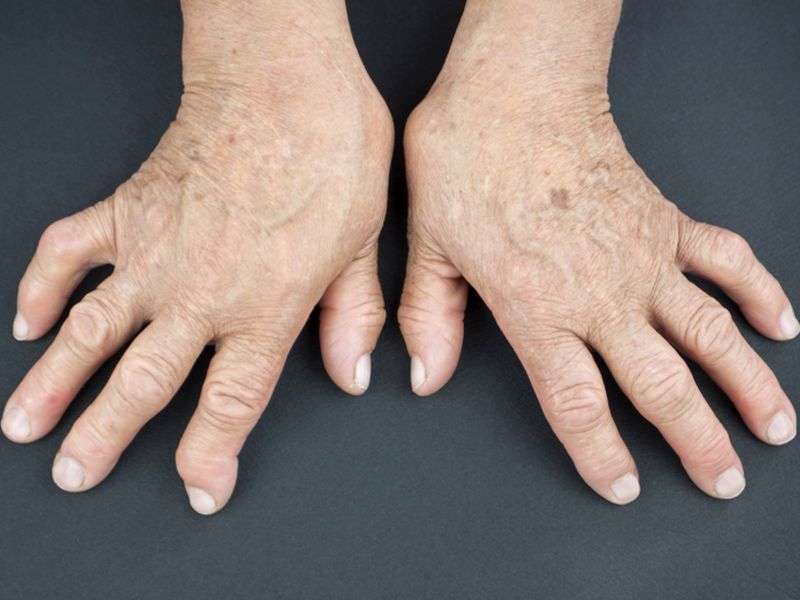
(HealthDay)—For patients with rheumatoid arthritis who have had an inadequate response to methotrexate, baricitinib is associated with significant clinical improvements, compared to either placebo or adalimumab, according to a study published in the Feb. 16 issue of the New England Journal of Medicine.
Peter C. Taylor, M.D., Ph.D., from University of Oxford in the United Kingdom, and colleagues conducted a 52-week, phase 3, double-blind controlled trial, in which 1,307 patients with active rheumatoid arthritis who were receiving background therapy with methotrexate were randomly assigned to one of three regimens in a 3:3:2 ratio: placebo (switched to baricitinib after 24 weeks), 4 mg of baricitinib once daily, or 40 mg of adalimumab (an anti-tumor necrosis factor α monoclonal antibody) every other week.
Using the American College of Rheumatology criteria, the researchers found that more patients had a 20 percent improvement at week 12 with baricitinib versus placebo (70 versus 40 percent; P < 0.001) or adalimumab (70 versus 61 percent; P = 0.014). All major secondary objectives, including inhibition of radiographic progression of joint damage at week 24, were met with baricitinib versus placebo (mean change from baseline, 0.41 versus 0.90; P < 0.001). Compared with placebo, adverse events (including infections) were more frequent through week 24 with baricitinib and adalimumab. Cancers were reported in five patients (two taking baricitinib and three taking placebo). Reductions in neutrophil counts and increases in levels of creatinine and low-density lipoprotein cholesterol were noted with baricitinib.
"In patients with rheumatoid arthritis who had had an inadequate response to methotrexate, baricitinib was associated with significant clinical improvements as compared with placebo and adalimumab," the authors write.
The study was supported by Eli Lilly, the manufacturer of baricitinib, and Incyte.
More information: Abstract/Full Text (subscription or payment may be required)
Copyright © 2017 HealthDay. All rights reserved.