A magic hood for artificial heart pumps

Ten million people in Europe alone suffer from cardiac insufficiency, or a weak heart. One day, many of them may require a heart transplant. Artificial heart pumps are frequently used to bridge the wait for the transplant. However, these pumps also have their drawbacks. A project involving Empa offers a possible solution.
Artificial heart pumps can be tricky: blood clots may develop, causing a stroke, and the immune system may attack what it recognizes as foreign. In order to tackle this and other problems with artificial hearts, in 2011 University Medicine Zurich launched the Zurich Heart project in collaboration with the University Hospital Zurich, the University of Zurich and the ETH Zurich. Meanwhile, the consortium boasts more than 75 MDs, engineers, biologists and materials scientists. Zurich Heart aims to refine current heart pumps and find completely new, original solutions. The goal of the project is to develop a fully implantable artificial heart. Around 20 research groups in Switzerland and at the German Heart Institute in Berlin have pooled their outstanding expertise to realize this ambitious goal.
As a materials research institute, Empa was a logical partner to contribute innovative solutions. "We'd like to make an artificial heart pump that works in a similar way to the human heart and has its inner surface covered with the patient's own cells," explains Edoardo Mazza, Head of Empa's Mechanical Integrity of Energy Systems lab, professor at ETH Zurich and co-leader of Zurich Heart. Two teams from Empa, one from the field of biotechnology/biointerfaces, the other from the textiles sector, are working on this heart pump, which is "invisible" to the body's blood clotting and immune system.
Textiles and human tissue
But what have textiles got to do with human organs? More than you might think: after all, aren't we talking about human tissue, about muscle fibers that can tear? And aren't veins and arteries essentially nothing more than hollow fibers, through which our blood flows? "These days, textile development has nothing anymore to do with cotton T-shirts and the like," says René Rossi, Head of Empa's Biomimetic Membranes and Textiles lab. For him, a textile is when a one-dimensional material – a fiber – is turned into a two-dimensional entity. This might be fabric or tissue. "Theoretically, there are no bounds to the materials and properties," explains Rossi. "The fibers can be made of metal, wood or synthetic material, and used to produce textiles or entities that are malleable, elastic, lightweight etc."
One of the main problems of current heart pumps is the fact that blood can start coagulating when it comes into contact with them. Blood clots can form, which roam around the body and can cause strokes or embolisms. If the surface of the artificial heart pump is given a kind of coating that the body perceives as a "natural" environment, however, blood clots could – at least in theory – be prevented.
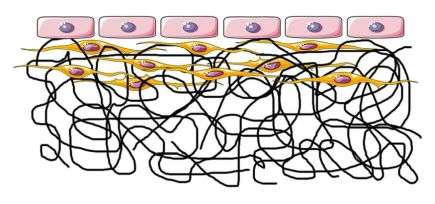
The inner surface of natural blood vessels is lined with a layer of endothelial cells, which control the exchange between the blood and body's tissues. Therefore, Empa scientists are now working on an ultra-thin lining made of sticky polymer fibers that are less than a micrometer thick. Living endothelial cells are placed on this "fabric", where they form a monolayer – as they do in all lymphatic and blood vessels. This kind of tissue surface could trick blood cells into thinking the pump is one of the body's own organs. For the endothelial cells to feel at home in the artificial tissue, however, they need to be able to effectively "cling onto" the lining. A simple lining made of polymer fibers is ill-suited.
Electrospinning and living cells
This is where Empa's electrospinning unit comes in. The technique is used to produce polymers, i.e. purely or-ganic and hybrid fibers measuring less than a micrometer in diameter, which makes novel membranes for use in medical engineering, catalysis and filter technology possible. An electric current is applied between a cannula, from which a polymer solution is squeezed, and a counter electrode – and this pulls threads. Thanks to the electric field, the threads twirl until they form a tissue-like membrane. Holding it in your hand, it feels like a wafer-thin, elastic cloth. "The membrane for the heart pump needs to be stable, firm and stretchable in all directions," says Giuseppino Fortunato from the Biomimetic Membranes and Textiles lab. "It really has to withstand a lot." After all, the heart beats around 100,000 times a day.
Muscle cells form the foundation
This mixed tissue made of fibers and cells can also be produced in the incubator. The Biointerface lab headed by Katharina Maniura is in charge of the cells, using smooth muscle cells that form a cellular structure on the hybrid membrane – as found in natural blood vessels. Endothelial cells are then supposed to settle on this "substructure".
The cells feel particularly at home if they find a substructure that reminds them of the body's own structures – more specifically, collagenous fibers such as from connective tissue. "We have to get muscle cells to produce collagen so the endothelial cells stick to it permanently," explains Maniura. "If the tissue is composed of two types of cells, they emit signals and thus communicate with each other. This also has the effect that the endothelial cells are stabilized on the surface and readily perform their natural functions." In order to render the electrospun fibers particularly attractive for the cells, the polymer fibers should be functionalized with cell adhesion peptides. The idea is for both the endothelial and the muscle fiber cells to be "presented" with their typical natural environment so the multi-layered structure lives as long as possible.
Verifying process in the bioreactor
Whether this also works in practice is being examined in a bioreactor. The material system, i.e. the synthetic elastomer pump wall developed by the Zurich Heart consortium, is exposed to "real" conditions together with Empa's cell-textile mixture.
The reactor recreates the situation in the human body, allows a cell culture liquid instead of blood to flow by, and simulates pulsations that mimic the movement of the heart muscle. This should show the researchers whether the "camouflaged" materials can withstand the heavy strain in the human body.
"We will conduct a study using the first prototypes of the biomimetic heart pumps before the end of the year. But it will be many years before they can be used clinically," says Mazza. The pumps each have to be "grown" individually using the patient's own cells. Eventually, this will involve taking cells from the patients' blood, vessels or fatty tissue. These will then be grown in the lab for two to three weeks before the heart pump with the endothelial layer can be implanted.
"The concept would be too slow for emergency operations," explains Mazza. Nonetheless, patients with a cardiac insufficiency could use the biomimetic pump to relieve the strain to such an extent that the heart is able to regenerate under its own steam – help it to help itself, as it were.