Molecular link between Parkinson's disease and prion diseases
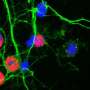
Parkinson's disease and prion diseases are very different as regards both origins and course. Nonetheless, a research group of SISSA, headed by Professor Giuseppe Legname, has discovered an unexpected and important link between the two pathologies. According to the study, recently published in the journal Scientific Reports, the pathologies are linked by the complex interaction between two proteins present in nerve cells: the so-called α-synuclein in its aggregated form, and the prion protein PrPC, the molecule responsible, in its altered version, for serious syndromes such as Creutzfeldt-Jakob disease.
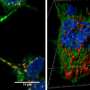
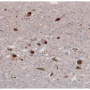
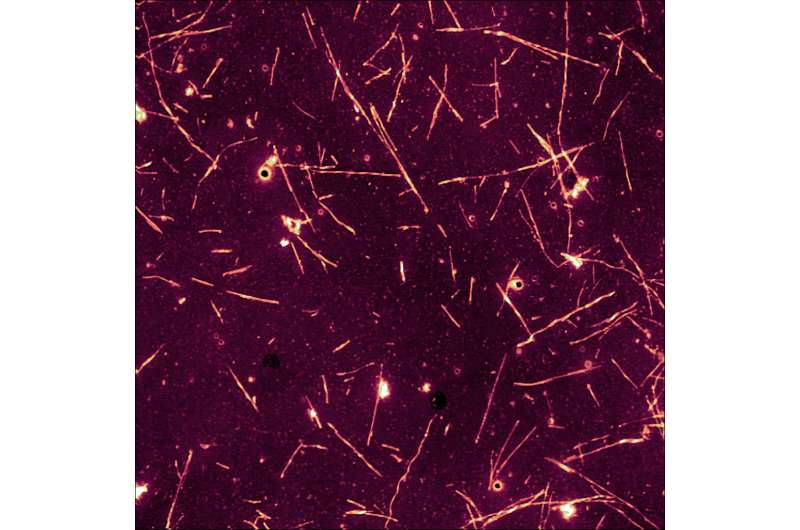
The presence of α-synuclein deposits in brain cells is typical of diseases technically called synucleinopathies, which includes Parkinson's disease, characterized by dementia with Lewy bodies and multiple system atrophy. However, the modalities according to which these aggregates form and spread were unknown until now. This researchers discovered that α-synuclein actually makes use of the action of the prion protein to spread and deposit in the brain. This seems to favour the formation of these deposits and their spread among brain cells.
Additionally, while the activity of the prion protein seems to support the development of synucleopathies, α-synuclein deposits seem to slacken the course of prion diseases. In fact, this research has proven that α-synuclein fibrils block the deposit of prions in nerve cells, thus preventing their replication. This surprising effect is corroborated by further evidence already found in the pathology—in fact, the course of the disease is slower in patients affected by prion diseases presenting α-synuclein deposits in nerve cells.
More information: Suzana Aulić et al, α-Synuclein Amyloids Hijack Prion Protein to Gain Cell Entry, Facilitate Cell-to-Cell Spreading and Block Prion Replication, Scientific Reports (2017). DOI: 10.1038/s41598-017-10236-x