Brain stimulation shows promise for Alzheimer's disease in early trial
Scientists in the US have found that electrically stimulating regions of the brain in three people with Alzheimer's disease was safe and appeared to show an effect on thinking skills and day-to-day tasks in two of the volunteers. The findings are published today in the scientific journal, Journal of Alzheimer's Disease.
There has been no new licensed treatment for Alzheimer's disease in over 15 years and in this early-stage study scientists looked to see if deep brain stimulation (DBS) improved symptoms in three people with Alzheimer's. DBS is similar to a heart pacemaker but for the brain instead of the heart and has widely been used as a surgical treatment for people living with Parkinson's disease. The device electrically stimulates brain regions responsible for decision making through probes implanted deep in the brain.
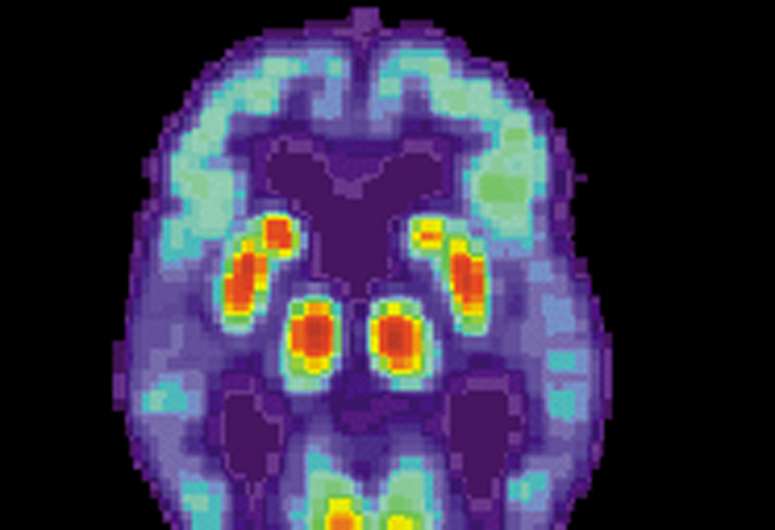
Three participants with Alzheimer's disease took part in the trial and carried out a series of memory and thinking tests. They were also scored on how well they carried out day-to-day activities, before receiving surgery to have the probes and device implanted. Participants also had brain PET scans taken throughout the trial period to assess how the brains energy patterns were affected.
After initial surgery to implant the device, the study volunteers received DBS for at least 18 months, and any side effects were addressed by adjusting the settings on the implant. Volunteers reported no serious side-effects by the end of the trial period.
Researchers matched each of the three trial participants to larger groups of people who were of a similar age and had similar memory and thinking abilities at the beginning of the study, to allow for comparisons to be around their performance. However, the comparison group did not have surgery or any dummy treatment.
After at least 18 months, two of the three participants showed less decline in memory and thinking compared to their matched groups. Scientists also looked at PET scans and found the same two participants showed increased brain activity in regions responsible for decision making and problem solving. Researchers also found that one of the three participants was better at performing daily tasks like preparing simple meals compared to the start of the trial after two years of stimulation.
Dr. Carol Routledge, Director of Research at Alzheimer's Research UK, said:
"Deep brain stimulation is already used as a treatment to reduce severe tremors in Parkinson's and it is encouraging to see this exploratory study in people with Alzheimer's disease. Previous research with deep brain stimulation in Alzheimer's has shown mixed results, but studies have not focused on brain regions responsible for decision making and problem solving before. While memory is a key problem in Alzheimer's, changes in thinking skills have an equally devastating impact so it's important that treatment approaches address these symptoms too.
"This small phase I trial is useful in demonstrating that this invasive treatment is safe and has no serious side effects in Alzheimer's, but the observed benefits in two of the patients must be treated with caution. The study did not compare against a dummy treatment and so while signs of benefit are worthy of follow-up, the full benefits and cost-effectiveness of this treatment need much more robust investigation in larger trials.
"There is a large amount of ongoing research to develop new drug treatments for dementia, but it's important that research also explores non-drug avenues of treatment. With 50 million people in the world living with dementia today, it is vital we continue to invest in a broad range of approaches to improve people's lives."