How the germ behind flesh-eating disease hijacks neurons to avoid immune destruction
The germ that causes garden-variety strep throat gets most of its bad rap for the misery of swollen tonsils and missed school days, but this wily pathogen has a lesser known, darker side.
The bacterium, known as Streptococcus pyogenes, is also the leading cause of serious flesh-eating disease, known as necrotizing fasciitis, which occurs in as many as 1,200 people each year in the United States and in 200,000 people worldwide. Although rare, the infection, which burrows deep under the skin and eats into connective tissue and muscle, is notoriously hard to diagnose and treat promptly and can rapidly become fatal.
What enables this bacterium to foil the body's defenses and wreak such massive tissue damage?
So far, the answer has remained elusive, but new research, led by Harvard Medical School scientists and conducted in mice, provides intriguing insight into the tactics used by S. pyogenes and points to several new ways to contain it.
Findings of the federally funded study, published May 10 in Cell, reveal that, to ensure its survival, the germ hijacks neurons and exploits the normal communication that occurs between the nervous and immune systems during injury or infection.
The study also suggests two distinct approaches involving nerve modulation to avert disease and treat these infections in mice. If replicated successfully in larger animals and in humans, these treatments could be used to block the germ's dangerous moves, prevent widespread infections and halt disease progression.
"Necrotizing fasciitis is a devastating condition that remains extremely challenging to treat and has a mortality rate that's unacceptably high," said study senior investigator Isaac Chiu, assistant professor of microbiology and immunobiology at Harvard Medical School. "Our findings reveal a surprising new role of neurons in the development of this disease and point to promising countermeasures that warrant further exploration."
Flesh-eating infections usually start with intense localized pain that is swiftly followed by the rapid death of connective and muscle tissue, rendering prompt treatment a challenge. Necrotizing fasciitis claims the lives of as many as 3 in 10 people who get the disease, according to research. Those who survive typically need radical, sometimes disfiguring, surgery to excise diseased muscle and connective tissue. In some cases, the disease causes such extensive damage that it may lead to limb amputation.
"We hope our findings can lead to new treatments for a condition that remains rare but can inflict significant damage and even death," said study first author Felipe Pinho Ribeiro, a post-doctoral researcher in the Chiu lab.
When the body is injured, the nervous system springs into action. Nerve cells send two separate memos. One of them goes to the brain, telling it that something is wrong, triggering the sensation of pain. The other goes to the immune system, telling it to keep away. This "stay away" signal plays an important protective role. In the setting of tissue injury or trauma, an overactive immune system can inflict serious collateral damage on healthy tissue when it deploys an army of disease-fighting cells. To prevent this sort of immune mayhem, neurons can send a chemical missive telling the immune system to keep its attack dogs in check.
"We believe this is the body's way of ensuring the delicate balance between alerting the body of brewing trouble while at the same time keeping overzealous immune cells at bay," Pinho Ribeiro said.
It is precisely this normal part of the crosstalk between the two systems that the germ behind flesh-eating disease exploits to avoid immune destruction, the researchers found.
Chiu's interest in the interplay between nerve signaling and immunity in the context of necrotizing fasciitis was ignited a few years ago during discussions with Michael Wessels, professor of pediatrics, medicine and microbiology and immunobiology at Harvard Medical School and chief of infectious diseases at Boston Children's Hospital.
Wessels shared with Chiu his interest in a rather intriguing feature of flesh-eating disease: The patients Wessels and others saw had intense "pain out of proportion" before any visible signs of the disease had emerged.
Chiu began wondering whether pain was more than an innocent indicator of brewing trouble. Could this normal pain response somehow be implicated in disease development?
That question prompted Chiu and colleagues to focus on neurons as a possible site of the action.
In the current study, the team's first order of business was to create a high-fidelity animal model of human disease. To do so, Chiu and colleagues injected mice with bacterial strains that came from patients with invasive strep infections, including necrotizing fasciitis.
An initial set of experiments identified the bacterial toxin streptolysin S—already known to kill red blood cells when secreted by the strep bacterium—as the key catalyst triggering pain and the ensuing immune-silencing cascade inside neurons. Indeed, mice infected with bacteria genetically modified to lack the toxin showed no signs of pain with infection, nor did they develop invasive disease. When re-infected with mutant germs reengineered to make the toxin, the animals developed full-blown disease. When researchers gave mice a neutralizing antibody that inactivated streptolysin S, the animals showed fewer symptoms suggestive of pain, indicating this toxin is, indeed, a key driver of pain.
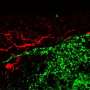
Further experiments showed that once in contact with neurons, streptolysin S prompts them to dispatch a pain signal to the brain alerting it that something is wrong, while at the same time inducing them to release a nerve chemical, the neurotransmitter CGRP (calcitonin gene-related peptide), to keep the immune system at bay. Experiments showed that CGRP actively interferes with the body's immune defenses in two ways. First, it impedes the body's ability to summon disease-fighting immune cells called neutrophils to the site of infection. Second, it inhibits any neutrophils that manage to make their way to the epicenter of infection from releasing an enzyme that secretes a bleach-like, germ-killing substance.
"Effectively, this neuronal signal silences the alarm system that normally calls on the body's infection fighters to curb infection," Chiu said.
In the case of clean, uninfected wounds, this is precisely the right response needed to prevent tissue-damaging inflammation caused by an overactive immune system, Chiu said. In this instance, however, it allows the strep bacterium to continue unhindered on its tissue-damaging march.
The absence of inflammation-inducing neutrophils at the site infection, the researchers say, may explain why, in the early stages of necrotizing fasciitis, patients tend to experience intense pain but without the heavy swelling, redness and overall inflammation that develop when bacteria-gobbling neutrophils rush to the wound.
To confirm neurons as the actual site of all action, the researchers used chemical and genetic methods to silence pain fibers in a subgroup of mice. When infected with the disease-causing bacterium, these animals controlled their infections better than mice with intact neurons.
Next, the scientists injected a group of mice with the nerve-blocking substance botulinum neurotoxin A—the active ingredient in cosmetic products that temporarily remove facial wrinkles.
A week after getting the nerve-block injections, animals were infected with the disease-causing bacterium. Compared with mice that didn't get nerve-block injections, pretreated mice developed only minimal wounds that never progressed to full-blown disease. In another experiment, researchers administered botulinum injections not before but immediately after infection. Again, the nerve-block injections controlled the spread of infection, resulting only in small localized lesions. A third group of mice got the nerve-block injections with some delay—two days after infection and once they had already developed a wound. The injection essentially stopped infection in its tracks and averted further damage to muscle and soft tissue.
In a final set of experiments, the researchers blocked CGRP's immune-suppressing activity by treating mice either with an injectable or ingestible form of the CGRP-blocking molecules. This treatment rendered immune cells deaf to the "stop" signal sent by the neurons and successfully prevented the spread of necrotizing fasciitis in mice infected with the disease-causing bacterium.
Botulinum injections are already approved by the FDA for a variety of cosmetic and therapeutic uses, including the treatment of migraine. CGRP blockers are nearing FDA approval as migraine treatment.
The results indicate that both the injectable nerve-block approach and antibody or other drug-based treatments against CGRP hold therapeutic promise for necrotizing fasciitis, the team said.
"Our findings provide a striking example of how closely intertwined the nervous and immune systems are and how intricate their interaction can be in the setting of infection," Chiu said. "Our study also underscores the therapeutic potential of modulating one system to affect the other as a way to treat infection."