New understanding of the way chikungunya virus protects mice against malaria could lead to improved patient care
Chikungunya virus (CHIKV) infection may reduce the severity of malaria, according to a discovery by A*STAR scientists, which could lead to the development of new malaria treatments.
Spread by different mosquito species in many tropical countries, the two infections commonly occur in tandem, recent studies have found. Cases of CHIKV have risen significantly in the last decade and co-infection with malaria is frequently misdiagnosed as malaria only.
As diagnosis methods improve, a better understanding of the effects of co-infection will help treatment options. Previous research by Laurent Rénia of the A*STAR Singapore Immunology Network (SIgN) has shown that 70-80 per cent of mice infected with the parasite Plasmodium berghei, widely used by scientists as a model of human malaria, die within six to 12 days.
In a new study, a team led by Rénia and Lisa Ng, also of SIgN, showed similar results for mice infected with P. Berghei, both four days before and after being also infected with CHIKV. However, among another group simultaneously infected with both, close to 75 per cent survived for at least 26 days.
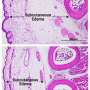
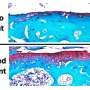
The researchers found that mice infected with both pathogens had lower levels of P. berghei in their brains and less damage to the blood brain barrier than those infected only with the malaria-causing parasite.
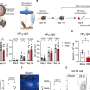
CD8+ T cells, a type of white blood cell, play important roles in the body's defenses against pathogens including malaria, using various methods to attack and kill infected or malignant cells. The team showed that levels of CD8+ T cells were lower in the brains of co-infected mice than in those of mice with malaria only, while remaining similar in the spleen.
Rénia and Ng went on to discover the complex chain of molecular events behind the protective effects of co-infection. Namely CD4+ T cells, which regulate immune responses, secrete a protein called interferon gamma, causing changes in levels of proteins involved in cell signaling and trafficking. This, in turn, reduces migration of CD8+ T cells to the brain, which, in the case of infection with malaria alone, can cause severe neurological damage.
This improved understanding of the mechanisms underlying the protective effect of CHIKV on malaria in mouse models could provide the basis for novel human therapies. "Targeting this same pathway to prevent or reduce CD8+ T cell migration to the brain could offer a new way to treat malaria," said Ng.
More information: Teck‐Hui Teo et al. Co‐infection with Chikungunya virus alters trafficking of pathogenic CD8 + T cells into the brain and prevents Plasmodium ‐induced neuropathology, EMBO Molecular Medicine (2017). DOI: 10.15252/emmm.201707885