Researchers show how the spatial organization of a nerve cell influences how its processes degenerate
During the development of the human nervous system, billions of nerve cells connect in order to communicate with each another. To this end, they use axons and dendrites. Wrongly matched processes, or those which are no longer needed, degenerate in the course of development. This degeneration is highly specific, as it affects only the part of a process that is no longer needed. But how do nerve cells determine which part of the axons or dendrites to degenerate and which to preserve?
Researchers at the University of Münster (Germany) now report a correlation between the spatial organization of a nerve cell and the degeneration of its processes. Using fruit flies as a model system, they discovered that the specific arrangement of components of the cytoskeleton influences the direction in which the dendrites of a certain type of nerve cells degenerate during development. "This is the indication of how the degenerative process in neuronal processes is regulated spatially," says Dr. Sebastian Rumpf, a junior researcher group leader at the Cluster of Excellence and head of the study. In the long term, the results will help scientists to gain a better understanding of how the nervous system develops and how neural pathways connect with each other correctly in the brain. The study has been published in the journal Development.
"Pruning" is the term describing the degeneration of nerve cell processes. The mechanisms underlying pruning are the major focus of a team of biologists headed by Sebastian Rumpf who are studying this process in the fruit fly Drosophila melanogaster. The team had discovered earlier that components in the cytoskeleton—the tubular microtubules—perform a key role in the pruning process. "They fall apart, and as a result, the dendrites then also degenerate. This process is initiated by a signaling protein that we discovered last year," says Dr. Svende Herzmann, the lead author of both the earlier and the current studies. "What we then wanted to know was which spatial aspects play a role within the cell during this process."
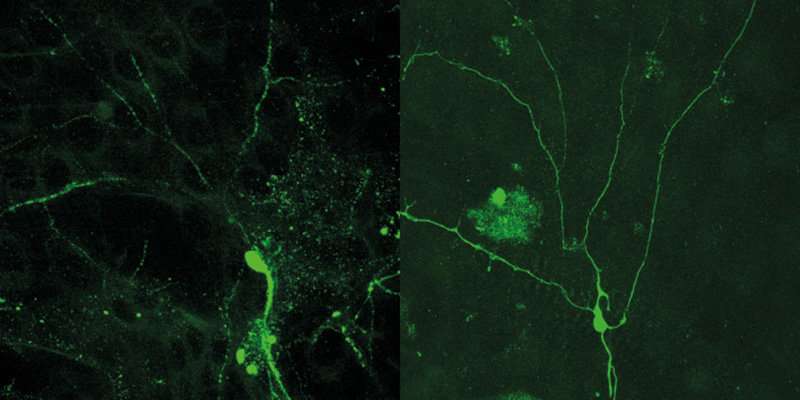
To address this question, the researchers marked the microtubules in the dendrites of fruit fly larvae with fluorescent dyes and visualized them by confocal microscopy. They then tracked the behaviour of the microtubules over time. "What we observed was that the microtubules are particularly sensitive at the branching points of the dendrites close to the cell body of the nerve cells and that they disintegrate there first," says Sebastian Rumpf. These gaps spread out into the lateral branches—correlating with the direction of the breakdown of the dendrites themselves.
"We asked ourselves whether the spatial orientation of the microtubules might be important for this," says Svende Herzmann. Microtubules have an inherent polarity: each "tube" has a so-called plus end and a minus end. It was known from earlier studies that the plus ends in the dendrites are always oriented towards the cell body. In axons, it is exactly the other way round—and in these processes, degeneration proceeds from the tips to the branchpoints, the exact opposite orientation of the dendrites.
The researchers then used genetics to inactivate kinesin, an important motor protein, thus changing the uniform alignment of the microtubules in the dendrites. Remarkably, this specific manipulation prevented dendrite degeneration. "Thus, there is a correlation between the spatial organization of the cell and the pruning process," says Sebastian Rumpf. "This is an extremely exciting hypothesis which we will consolidate in further studies." As a next step, the researchers want to use high-resolution microscopy to take a more detailed look at the special processes at the dendrite branching points—which will enable them to find out more about the mechanisms which regulate the degeneration of the nerve cell processes.
More information: Svende Herzmann et al, Spatial regulation of microtubule disruption during dendrite pruning inDrosophila, Development (2018). DOI: 10.1242/dev.156950