Important signaling pathway in breast cancer revealed
Researchers at Kanazawa University report in Proceedings of the National Academy of Sciences (PNAS) that a particular signaling pathway in breast cancer tumors causes cancer cells to divide symmetrically, expanding the tumor. Inhibiting the pathway with drugs could become a strategy for eliminating the cancer cells.
In breast cancer, one of the most common cancers in women, tumors contain a small amount of so-called cancer stem-like cells (CSCs). Eliminating breast cancer stem-like cells in a targeted way is essential for developing successful therapies—conventional treatments, such as chemotherapy or radiotherapy followed by drug intake, do not target CSCs. A better understanding of the processes generating CSCs in breast cancer tumors is needed. Noriko Gotoh from Kanazawa University and colleagues have now uncovered a signaling pathway directly related to the proliferation of CSCs in breast cancer.
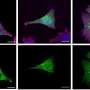
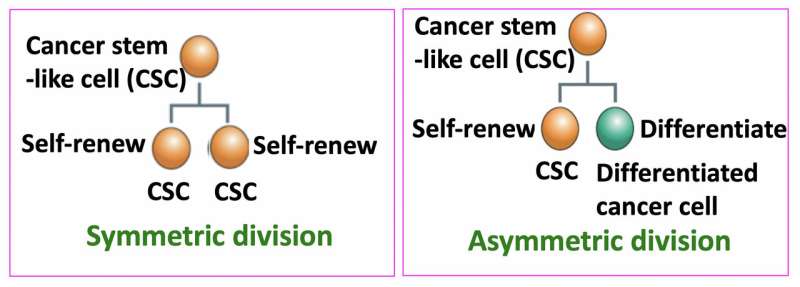
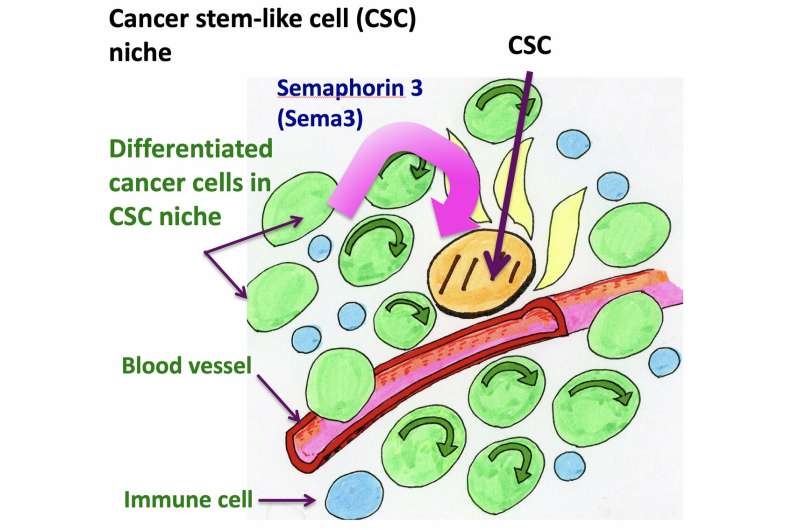
Stem cells can self-renew and differentiate. Two types of stem-cell division can occur: symmetric or asymmetric (Fig. 1). In symmetric division, two self-renewing stem cells are generated; in asymmetric division, only one stem cell and a differentiated cell are generated. It is now believed that malignant CSCs have a higher tendency to divide symmetrically, thus increasing the number of CSCs. Gotoh and colleagues looked at how the microenvironment of CSCs, called the CSC niche, causes and sustains an increased rate of symmetric division.
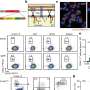
The researchers started from the observation that a particular gene encoding a type of cytokine known as Semaphorin 3 (Sema3) was one of the most highly expressed genes in the CSC niche (Fig. 2). (Cytokines are small proteins that, when released, affect the behavior of cells around them.) The production of Sema3 activates another protein, called MICAL3, the expression levels of which were also found to be high in the CSC niche.
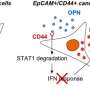
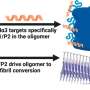
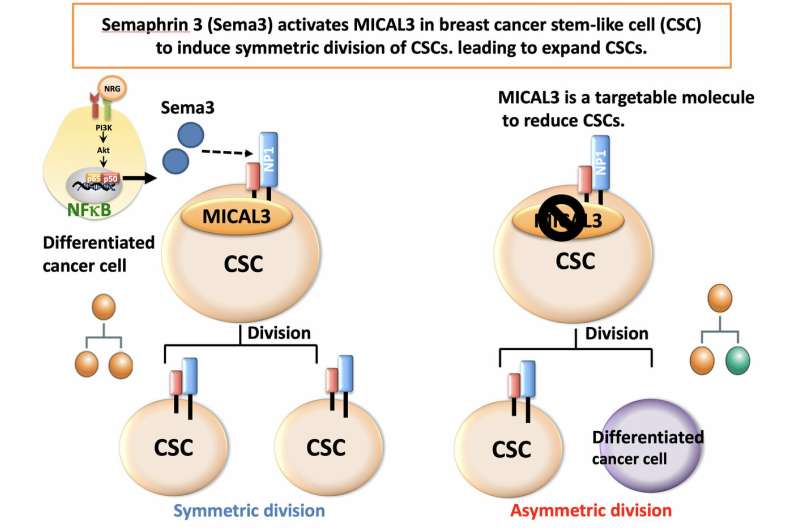
Via a series of experiments in vitro, Gotoh and colleagues were able to confirm the critical roles of Sema3 and MICAL3 in breast cancer tumor development. Specifically, MICAL3 was shown to be required for tumor sphere formation (tumorigenicity is associated with spherical cell shapes). The scientists showed that Sema3-stimulated MICAL3 triggered a whole sequence of biomolecular interactions (a signaling pathway), ultimately resulting in induced symmetric division of CSCs, and hence their proliferation, in breast cancer (Fig. 3).
Having established this important pathway is highly relevant for developing treatments for breast cancer, because, according to Gotoh and colleagues, "by inhibition of MICAL3... or knockdown of each component in the signaling pathway, the symmetric cell division may be inhibited, leading to a reduction of breast cancer stem-like cells."
More information: Kana Tominaga et al, Semaphorin signaling via MICAL3 induces symmetric cell division to expand breast cancer stem-like cells, Proceedings of the National Academy of Sciences (2018). DOI: 10.1073/pnas.1806851116