S1P molecule regulates dendritic cell localization and vascular integrity in lymph nodes
The trafficking of white blood cells called lymphocytes is crucial for controlling our immune response. Maintaining the integrity of high endothelial venules (HEVs)—the gateway for lymphocytes to move from the blood into lymph nodes (LNs)—is therefore crucial. This study describes for the first time how a molecule called sphingosine-1-phosphate (S1P) positively influences the function and integrity of HEVs. It also reveals how the activation of sphingosine-1-phosphate receptor-1 (S1PR1) signalling influences HEV interactions with dendritic cells (cells that carry messages between the innate and adaptive immune systems).
The researchers hope this work will inspire future research to better understand how endothelial cells (the cells that line the interior surface of blood vessels and lymphatic vessels) distinguish S1P-signalling from the signals generated by high levels of S1P in the blood, thereby ensuring the proper maintenance of our immunity and tolerance. Greater understanding of these processes could have implications for studying orchestration of anti-cancer immunity and autoimmune diseases such as rheumatoid arthritis.
Interview with Dr. Szandor Simmons
What question did you set out to answer in your study?
We intended to describe the influence of sphingosine-1-phosphate, a bioactive sphingolipid mediator, on the function and integrity of high-endothelial venules (HEVs), which are specialized postcapillary venules that facilitate lymphocyte immigration from peripheral blood circulation into e.g. lymph nodes.
Can you briefly summarize the key findings of your paper?
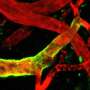
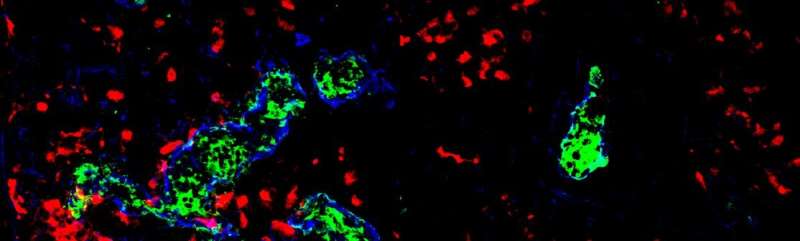
We found that HEVs, which were deficient for the S1P-transporter Spinster-homologue-2, were unable to attract dendritic cells in order to trigger LTβr-signaling through cell-cell contact with HEVs. We observed a comparable phenotype when HEVs were unable to signal through S1P-receptor-1. Therefore, CCL21 release from HEVs was reduced and DC-HEV interactions abrogated, which led to impairment of HEVs, reduced lymphocyte immigration into the lymph node parenchyma, and to the development of hypotrophic lymph nodes.
What is the impact of your work? How/why are your findings important for other scientists, and what could they mean for the everyday reader?
Lymphocyte trafficking is crucial for controlling adaptive immune responses. Hence, the maintenance of the integrity of high endothelial venules (HEVs), the gateway of lymphocytes from the blood into the lymph nodes (LNs), is a key issue. To our knowledge, so far, no studies have described the influence of sphingosine-1-phosphate on the function and integrity of HEVs. Furthermore, we describe how the autocrine activation of sphingosine-1-phosphate receptor-1 (S1PR1) signaling influences HEV and dendritic cell (DC) interactions.
Our work provides an explanation for the well documented organization of dendritic cells in the lymph node and hopefully will inspire future work to better understand how endothelial cells distinguish the autocrine S1P-signaling from signals generated by high levels of S1P in the blood. For maintenance of immunity and tolerance, the organs and tissues of the organism are connected by migrating lymphoid cells. Understanding lymphocyte migration is essential for many disorders and diseases.
More information: Szandor Simmons et al, High-endothelial cell-derived S1P regulates dendritic cell localization and vascular integrity in the lymph node, eLife (2019). DOI: 10.7554/eLife.41239