Treatment for 'low T' could someday come from a single skin cell, research shows
USC researchers have successfully grown human, testosterone-producing cells in the lab, paving the way to someday treat low testosterone with personalized replacement cells.
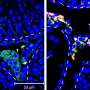
In today's Proceedings from the National Academy of Sciences, scientists describe how they transformed stem cells into functioning Leydig cells—the cells in the testes that produce the male sex hormone.
"Our study provides a way to generate possible transplantation materials for clinical therapies, as well as a path toward testing and developing new drugs," said Vassilios Papadopoulos, dean of the USC School of Pharmacy, who led the research.
Millions of men have low testosterone, or hypogonadism, which impacts mood, fertility, sexual function, obesity and bone density—and testosterone replacement therapy is a multibillion-dollar industry. Testosterone tapers off naturally with age but can also decrease suddenly due infections like mumps, or cancer treatment during childhood.
Testosterone replacement therapy—injected, taken orally or applied as a gel—reverses many of these symptoms.
"You feel better, you lose weight, erectile function returns," Papadopoulos said. "Men love testosterone."
However, treatment for "low T" is linked to side effects such as infertility, increased risk of prostate cancer and cardiovascular diseases. In addition, topical treatments can rub off on close contacts, inadvertently exposing others to the drug. A transplant of lab-grown testosterone-producing cells, perhaps injected into fatty tissue, could potentially bypass those side effects, researchers say.
Previous attempts to cultivate Leydig cells have come up short. In one study, the lab-grown cells produced cortisol, not testosterone, Papadopoulos said. Other experiments have involved stem cells from bone marrow or the umbilical cord; harvesting these cells is more labor-intensive and they do not multiply as well in the lab.
In Papadopoulos's experiment reported today, researchers started with stem cells called human-induced pluripotent stem cells, which come from human skin or blood and can be developed into any type of cell needed for treatment purposes.
On a hunch, Papadopoulos added human collagen to his soup of nutrients, genes and other ingredients needed to transform stem cells into Leydig cells. Collagen is a common growth matrix ingredient; previously, Papadopoulos used bovine or rat collagen, which are cheaper and mostly interchangeable with other forms of collagen, at least in early-stage experiments.
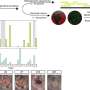
This time, the lab-grown Leydig cells produced testosterone—and the cells even looked the same as their naturally occurring counterparts under the microscope.
"It was none of the things we thought. We had tried different genes, chemicals, everything—nothing!" he said. "The human collagen was the secret sauce."
Next, Papadopoulos wants to test how well lab-grown Leydig cells function, and for how long, when they are transplanted into animal models of hypogonadism. He's also eager to compare Leydig cells cultivated from skin cells from men with and without hypogonadism, to better understand the condition.
Human transplantation of Leydig cells is at least "a few years away," he said.
More information: Lu Li el al., "Directing differentiation of human induced pluripotent stem cells toward androgen-producing Leydig cells rather than adrenal cells," PNAS (2019). www.pnas.org/cgi/doi/10.1073/pnas.1908207116