Brain organoids reveal glioblastoma origins
Glioblastomas are the most aggressive form of brain cancer—they grow and spread rapidly through the brain and are virtually impossible to eradicate, typically leading to death within one or two years of diagnosis. Scientists are constantly seeking more powerful targeted therapies, but so far without success—in part because glioblastomas are challenging to study in a laboratory setting.
"Glioblastomas are aggressive and tenacious in patients, but have always been very difficult to keep alive in the lab," said UC San Francisco postdoctoral researcher Aparna Bhaduri, Ph.D. "In previous attempts to study glioblastomas in mice, only 5 to 10 percent of human tumors survive transplantation into the animals, making us suspect that these tumors may differ in important ways from the ones that don't survive."
Now Bhaduri and Elizabeth Di Lullo, Ph.D., a fellow postdoctoral researcher in the laboratory of Arnold Kriegstein, MD, Ph.D., at the UCSF Eli and Edythe Broad Center for Regeneration Medicine and Stem Cell Research, have for the first time succeeded in keeping a diverse array of glioblastomas alive in the lab using brain organoids—balls of simple brain tissue grown from human stem cells.
"The tumors take to our organoid models nearly 100 percent of the time, meaning that we can finally study the full spectrum of glioblastomas in the lab," Di Lullo said.
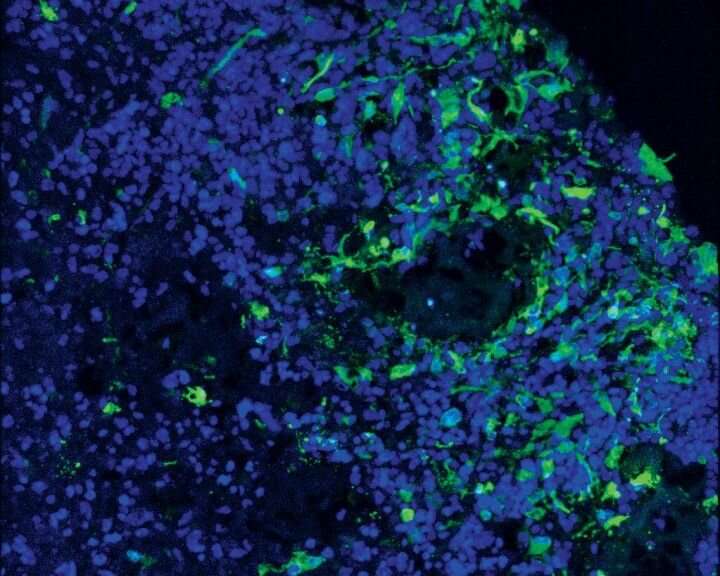
In a study published Jan. 2, 2020, in Cell Stem Cell, Bhaduri, Di Lullo, and colleagues first created an atlas of glioblastomas taken from surgical treatment of human patients, cataloguing dozens of distinct cell types and these cells' expression of distinctive patterns of genes. They then used organoids grown from human stem cells to model how these genetically identified cancer cell types behave in human brain tissue.
They discovered that glioblastomas do not appear to originate from a single cell type—what other researchers have called a "glioblastoma stem cell"—but instead arise from multiple kinds of seed cells, including one that looks and behaves very much like a neuronal stem cell Kriegstein's lab discovered a decade ago, called an outer radial glia (oRG) cell.
These oRG cells are much more numerous in humans and other primates compared to other mammals, and their rapid divisions and characteristic "jumping" migration during brain development may have contributed to the evolutionary expansion of the primate brain, Kriegstein and colleagues believe.
These cells normally disappear as the brain matures, but the new results suggest that they may reappear in glioblastomas and could help drive the tumors' ability to rapidly grow and spread through the brain. The fact that oRG cells are numerous in primates but lacking in mice could even explain why glioblastomas only take hold and spread in human brain tissue—or organoids—but have trouble surviving in mice, the authors say.
"Our study raises a number of important questions for the field, including how a stem cell present only in the developing brain re-emerges in an adult tumor," said Kriegstein, a professor of neurology and John G. Bowes Distinguished Professor in Stem Cell and Tissue Biology, who also directs the Broad stem cell center at UCSF. "Fortunately, Aparna and Elizabeth have now developed a model system in which we can begin to find the answers."
"This research combines numerous complimentary techniques to provide comprehensive evidence that glioblastomas revive stem cell programs from early brain development, including migratory behavior that may to contribute to their rapid spread," added co-author David Raleigh, MD, Ph.D., a UCSF Health radiation oncologist and Helen Diller Family Comprehensive Cancer Center physician-scientist specializing in brain tumor biology. "This gives me confidence that the findings are not only scientifically robust but also will translate to human patients, with the potential to lead to new targeted therapies."
More information: Aparna Bhaduri et al. Outer Radial Glia-like Cancer Stem Cells Contribute to Heterogeneity of Glioblastoma, Cell Stem Cell (2020). DOI: 10.1016/j.stem.2019.11.015