New study sheds fresh light on the genetic mechanisms involved in sepsis
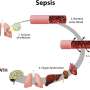
Researchers from the University of Granada (UGR) have generated new data on the genetic mechanisms associated with sepsis, an exaggerated inflammatory response to infection. Sepsis is the No. 1 cause of death in all hospital intensive care units (ICUs) worldwide.
The research group, led by Professor Darío Acuña-Castroviejo, recently published its findings in Shock. Their study analyzed changes in the expression of the so-called 'clock genes' (those that regulate the function of every cell in the body) of sepsis patients and the connection between these changes and the degree of inflammatory response, oxidative damage and urinary excretion of 6-sulfatoxymelatonin—the major melatonin metabolite and mediator of clock genes.
Sepsis leads to the formation of numerous pro-inflammatory molecules, such as inducible NO synthase, which produces large amounts of nitric oxide. This is an extremely powerful vasodilator that, together with other molecules, produces a drop in blood pressure and decreased blood flow to the tissues. These two responses constitute the basis of the hypovolemic shock and multi-organ failure that sepsis patients develop, culminating in their death.
In Spain, there are about 180,000 cases of sepsis per year, with a mortality rate of 25% among ICU patients worldwide. Treatment consists of antibiotics to halt the infection, as well as interventions to curb the systemic inflammatory response and restore normal blood pressure. However, there are currently no specific treatments for sepsis to control this inflammatory response or the resulting multi-organ failure.
Clock genes form part of the central system known as the circadian clock, which regulates the body's rhythmic functions. From the sleep/wake rhythm to those of brain neurotransmitters, hormones and metabolism, all circadian rhythms are under the control of this clock.
The activity of the immune system is another rhythmic function controlled by clock genes, which act as a necessary brake to this activity. When there is an abnormality in the expression of these clock genes, the body's innate immunity can go into overdrive. In turn, some of the proinflammatory cytokines produced by the inflammatory activation alter the clock genes themselves, setting in motion a vicious circle that fosters an exaggerated inflammatory response
Expression of clock genes in sepsis patients
This is the hypothesis analyzed in the published study. The research involved determining the expression of clock genes in the blood of sepsis patients, and comparing it to that of patients who were not suffering sepsis but who were hospitalized in the same ICU and were therefore under the same environmental conditions.
The results indicated that the degree of abnormality of the clock-gene expression is directly linked to the severity of the inflammatory response. When sepsis patients recovered and the inflammation was reduced, the expression of the clock genes was also stabilized. The alteration of the clock genes was also found to be related to the degree of oxidative stress produced by the inflammatory response, as well as to the biochemical markers of sepsis, such as procalcitonin.
Furthermore, melatonin production increased directly in line with inflammation, being higher in sepsis patients than in non-sepsis patients. The melatonin was negatively associated with oxidative stress indices and inflammation markers, indicating that its production was due to a compensatory response of the body to combat the inflammatory process.
The results of the study work demonstrate the role played by abnormality of the clock genes in the inflammatory response of sepsis, and indicated that melatonin is associated with an improvement in the evolution of this response and in patient outcomes.
More information: Daily Changes in the Expression of Clock Genes in Sepsis and Their Relation with Sepsis Outcome and Urinary Excretion of 6-Sulfatoximelatonin'. Shock, 2019; DOI: 10.1097/SHK.000000000000143.