Novel cell-based cancer immunotherapy shows promise in early studies
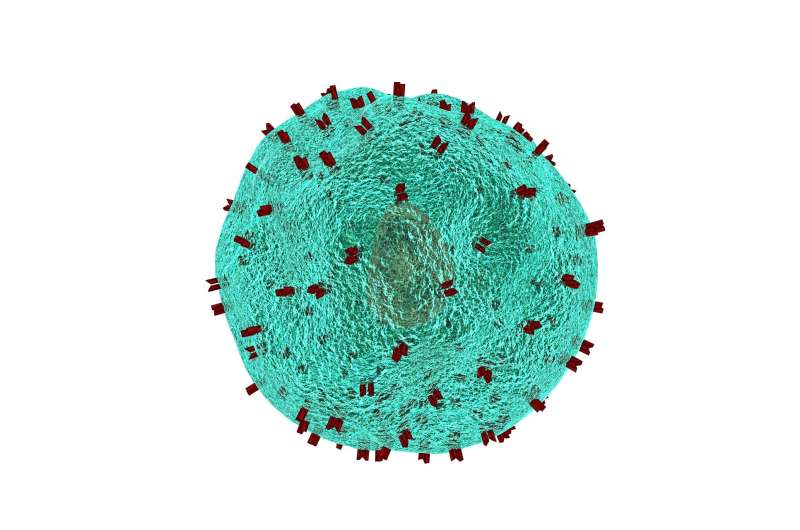
Scientists have developed a new immunotherapy that eradicates solid tumours in mice without adverse side effects, according to a new study published today in eLife.
The newly developed chimeric antigen receptor-T cell (CAR-T cell therapy) could soon be tested in clinical trials. In addition, the researchers used a new mouse model that could be used to test the safety, efficacy and mechanisms of CAR-T cell treatments for patients with solid cancers in future.
In CAR-T cell therapy, immune cells called T cells are collected from a blood sample of patients and reprogrammed with CAR molecules that recognise cancer-specific antigens expressed on cancer cells. When the CAR-T cells are given back to patients, they not only kill the antigen-expressing cancer cells directly but also switch on the immune system to fight the tumour.
"One of the challenges with developing CAR-T cells is that, sometimes, the CAR-T cells work against tumour antigens that are also present in lower amounts on normal cells, causing serious side effects which we only find out about in clinical trials," explains co-corresponding author Tomonori Yaguchi, Visiting Assistant Professor in the Division of Cellular Signaling, Institute for Advanced Medical Research, Keio University School of Medicine, Japan. "We proposed that if human CAR-T cells can cross-react with the mouse antigens and detect antigens found on normal mouse cells, we could test human CAR-T cells in mouse tumour models. This would allow more robust tests of both safety and effectiveness before the treatments reach clinical testing."
The team developed a CAR-T cell treatment targeting an antigen called glycipan-1 (GPC1). This antigen is found in large amounts on several types of human tumour cells, and also exists in low amounts on normal human and mouse cells. When the scientists tested CAR-T cells on mice bearing mouse tumours they found that the CAR-T cells effectively inhibited tumour growth without causing adverse side effects. In fact, for one of the mouse tumours, four out of five of the mice receiving CAR-T cell treatment remained completely tumour-free for at least 100 days. The team also found that the CAR-T cells enhanced immune responses against other tumour antigens than GPC1.
One of the most important new immunotherapy treatments for cancer are drugs called immune checkpoint inhibitors. These work by taking the brake off immune cells so that they can destroy cancer cells. When the team combined the CAR-T cells with a checkpoint inhibitory drug that blocks the activity of the PD1-protein found on T cells, this further enhanced the anti-tumour effects of CAR-T cell treatment, even though the checkpoint inhibitor alone had no effect on its own. This suggests that using CAR-T cells targeting GPC1 alongside a checkpoint inhibitor could be an effective combination treatment for cancer.
"We have generated CAR-T cells targeting GPC1 in both humans and mice and shown their effectiveness in mouse solid tumour models," concludes senior author Yutaka Kawakami, Professor in the Division of Cellular Signaling, Institute for Advanced Medical Research, Keio University School of Medicine. "By establishing a new type of model, we were able to test both the effectiveness, safety and anti-tumour mechanisms of CAR-T cells, showing the importance of choosing the most appropriate models for evaluating these novel types of cancer treatment."
More information: Daiki Kato et al, GPC1 specific CAR-T cells eradicate established solid tumor without adverse effects and synergize with anti-PD-1 Ab, eLife (2020). DOI: 10.7554/eLife.49392