Probing the genes that organize early brain development

When brains begin developing, there are a lot of moving parts—and when mutations happen in early neurodevelopment, it can lead to disorders like macrocephaly and autism. But scientists don't know much about the ways that development goes askew, particularly in humans.
That's why Wei Zhang, a postdoctoral scholar and research associate at USC's Center for Craniofacial Molecular Biology, became interested in how specific genes can impact early brain development.
From a previous study, Zhang and his colleagues had already honed in on a particular gene—RAB39b. They scoured the scientific literature to understand more about this gene and what it does.
RAB39b was reported as an autism spectrum disorder (ASD) risk gene, linked to the X chromosome, Zhang said. When this gene is mutated, it can lead to macrocephaly, ASD, intellectual disability, epilepsy and early onset Parkinson's disease. "However, the functional study of RAB39b is very limited, and the mechanism of RAB39b mutation leading to macrocephaly and ASD is not clear," he added. The researchers were intrigued and decided to investigate.
Macrocephaly/ASD belongs to a subset of autism. The brain surface area of a baby who will develop macrocephaly/autism hyper-expands in the weeks directly before and after birth, and then continues to grow larger than normal after birth, followed by the emergence of autistic social deficits. That makes the study of brain development critical for exploring the mechanism of ASD, Zhang said.
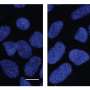
His lab already had a system developed to study brain development using a mouse model and human brain organoids—miniature brains grown artificially in the lab. These organoids emerged a decade ago as pea-sized blobs, grown from human stem cells that self-organize into brain-like structures with electrically active neurons.
They've been used to make discoveries about Zika-caused microcephaly, schizophrenia and even to test new drugs.
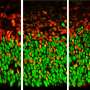
Using those two models, Zhang and his colleagues deleted the RAB39B gene in both the mice and the human brain organoid. The mice showed signs of macrocephaly and autism-like behaviors, and the human brain organoids were enlarged and impaired with the removal of the gene.
Zhang said this shows how a mutation in this gene disrupts growth and differentiation of so-called neural progenitor cells, which go on to become different brain cell types.
Ultimately, that can contribute to macrocephaly and autistic-like behaviors. The research was directed and performed in the laboratory of Jianfu Chen, an assistant professor of USC, and was published last month in the journal Genes and Development. Associate Dean of Research Yang Chai Ph.D. '91, DDS '96 also contributed to the research.
Approximately 1 in 59 children has been identified with autism spectrum disorder, according to estimates from the Centers for Disease Control's Autism and Developmental Disabilities Monitoring Network. Zhang said that this study can provide new insights into neurodevelopmental dysregulation and common pathways associated with macrocephaly and autism disorders across species.
Now that it's clear this gene has an important role in regulating new brain cell growth, Zhang and his colleagues hope to investigate RAB39b's role in regulating neuron activity-dependent signaling networks, for further exploring the mechanism of autism spectrum disorder.
More information: Wei Zhang et al. Cerebral organoid and mouse models reveal a RAB39b–PI3K–mTOR pathway-dependent dysregulation of cortical development leading to macrocephaly/autism phenotypes, Genes & Development (2020). DOI: 10.1101/gad.332494.119