New immunotherapeutic approach takes aim at cancer's enzyme shield
Immunotherapies have transformed the landscape of cancer treatment by allowing physicians to alter or augment patients' immune response to better attack malignant tumors. But some tumors—known as "cold tumors"—fly under the immune system's radar. Checkpoint inhibitors, which are the basis of many immunotherapies, don't work well against them. A team of investigators, led by researchers from Brigham and Women's Hospital, have focused on a protein called SerpinB9 (Sb9) with a potential role in cancer cells that has been underappreciated, but which could open the door to a new immunotherapy approach. The team observed that in a range of mouse models, inhibiting Sb9 with a small molecule reduced tumor growth both by weakening the tumor's defense mechanisms and by triggering cell death in the tumors themselves. The findings are published in Cell.
"In this study, we showed proof of concept using a small molecule that is designed to kill the cancer using its own lytic enzyme machinery," said corresponding author Reza Abdi, MD, of the Division of Renal Medicine at the Brigham. "Immunotherapies like monoclonal antibodies or checkpoint inhibitors are promising, heavily studied strategies, but antibodies are very hard to engineer and can also pose toxic effects to patients. A small molecule that inhibits the function of Sb9 could be simpler to develop, and potentially be more effective."
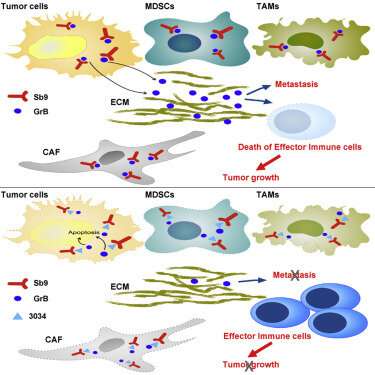
The researchers had known that in normal immune cells, Sb9 acts as a shield against the cells' own destructive enzymes, called granzyme B (GrB) enzymes. GrB enzymes are secreted to attack invading cells. However, the presence of Sb9 and GrB in cancer cells was not widely known. When the researchers examined a variety of human and mouse tumors, they saw heavy expression of Sb9, which could allow the tumor to resist attacks from GrB.
Using gene-editing CRISPR-Cas9 technology, the researchers engineered tumors that lack Sb9 and found that these tumors grew at a slower rate in mice. But they also observed that Sb9 was expressed in cancer-associated fibroblasts and immunosuppressive cells surrounding the tumor, which promote the growth of the cancer by weakening immune responses leveled against it.
"The initial findings showed that the tumor without the Sb9 protein grows slower. However, when we implanted knocked out Sb9 tumors in mice which lack Sb9, we observed a more notable reduction in tumor size," Abdi said. "These results suggested that if we could come up with a drug that systemically inhibits this protein in the tumor and in the cells of the host, we could get a synergistic benefit by simultaneously targeting various pathogenic arms of tumor formation, including the tumor, cancer-associated fibroblasts, and immunosuppressive cells."
The researchers developed a specific, small-molecule inhibitor that binds to Sb9 and inhibits its function in mice. Notably, the small molecule was effective in suppressing several murine models of solid tumors.
Abdi acknowledged that a significant amount of work still needs to be done to further optimize the binding kinetics of the small-molecule inhibitors of Sb9 and to determine the structural basis of interactions, and that rigorous toxicity testing must be completed before a drug can be taken to clinic.
"This protein could be extremely important for future cancer therapies, and the research community might have a better way to target this protein," Abdi said. "At the end of the day, we are excited to be amongst the very first to make a drug for this new target and show its potential as a novel approach to cancer therapy."
More information: Liwei Jiang et al, Direct Tumor Killing and Immunotherapy through Anti-SerpinB9 Therapy, Cell (2020). DOI: 10.1016/j.cell.2020.10.045

















