14-3-3sigma gene acts as a tumor suppressor in intestinal cancers
LMU researchers have identified the 14-3-3sigma gene as an important suppressor of carcinogenesis in the gastrointestinal tract.
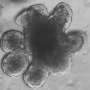
Intestinal cancers, also known as colorectal cancer, are among the most prevalent forms of malignancy worldwide. If detected early enough, tumors can be surgically excised. However, as cancer growth progresses, cells may escape from the primary tumor, which can then establish metastatic tumors in other organs. Once such satellite tumors have formed, survival rates fall significantly. Formation of the initial tumor can be triggered by mutations in any of a number of genes. Together with postdocs Markus Winter and Matja Rokavec, Professor Heiko Hermeking at LMU's Institute of Pathology has now discovered that a gene called 14-3-3sigma functions as a tumor suppressor in gastrointestinal cancers. Using the mouse as an experimental model, the team demonstrated that inactivation of the 14-3-3sigma gene enhance progression of gastrointestinal cancers and consequently shortens the lifespan of mice.
In earlier work with colon carcinoma cell lines, Hermeking and his colleagues had shown that a protein known as p53, which plays a central role in the suppression of many types of tumors, activates the 14-3-3sigma gene, and that its protein product inhibits passage through the cell cycle. This brake on cell division gives cells more time to repair adventitious or environmentally caused DNA damage, and therefore reduces the risk of uncontrolled cell proliferation. Mutations that inactivate p53 are found in more than half of all tumors. "But it was not clear whether the 14-3-3sigma gene itself plays an important role in the suppression of tumorigenesis in the intestine," says Hermeking.
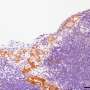
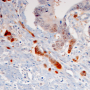
In a project funded by the German Cancer Research Fund (Deutsche Krebshilfe), his team set out to clarify this issue by analyzing patterns of gene expression in several thousands of tumor samples isolated from patients with colorectal cancer. The data revealed that the expression of 14-3-3sigma is indeed significantly downregulated in these tumors. In addition, this effect was particularly pronounced in metastatic tumors, and the level of expression of the gene showed a negative correlation with the degree of cancer progression. Furthermore, patients with lower levels of expression of the 14-3-3sigma gene were found to have a worse prognosis than those with higher expression—and this finding was independent of whether or not the tumors also contained an inactivating mutation in the p53 gene.
To further explore the significance of 14-3-3sigma in intestinal carcinogenesis, the LMU researchers specifically inactivated the gene in a preclinical mouse model of intestinal cancer. Loss of the gene's function was found to stimulate the formation and growth of tumors in the gastrointestinal tract, and ultimately reduced survival times. Subsequent studies showed that, in the absence of the 14-3-3sigma gene, signaling pathways which are regulated by transcription factors that have been implicated in the formation of metastases are selectively induced. Interestingly, the 14-3-3sigma protein is known to bind and negatively regulate these transcription factors. "Taken together, our results demonstrate that 14-3-3sigma is not only one of the many target genes that are controlled by p53 and mediate its tumor-suppressing function, but also has tumor-suppressive functions on its own in the gastrointestinal tract," says Hermeking.
The authors of the study suggest that detection of decreased expression of the 14-3-3sigma gene in resected primary colon carcinomas could serve as a prognostic indicator in patient populations. "Moreover, the signaling pathways that are triggered upon its inactivation are potential targets for therapeutic interventions," Hermeking adds.
More information: Markus Winter et al, 14-3-3σ Functions as an Intestinal Tumor Suppressor, Cancer Research (2021). DOI: 10.1158/0008-5472.CAN-20-4192