Derivative of vitamin A enhances the tumor-killing effectiveness of radiotherapy

Radiotherapy is a crucial component in cancer treatment, used in 50 to 60 percent of patients with cancer. It is traditionally used for localized cancers—such as head and neck, cervical, prostate, lung and brain cancers—with varying degrees of success.
A University of Chicago Medicine Comprehensive Cancer Center researcher-led team has discovered that combining radiotherapy with all-trans retinoic acid (ATRA) significantly inhibits the growth of not only locally irradiated tumors, but also distal tumors not treated with radiation. The combination treatment of radiation and ATRA modulates the tumor microenvironment and enhances the effects of radiation on both the local and systemic levels.
"Our group is the first to combine ATRA with radiation to treat solid tumors in animal models," said Ralph Weichselbaum, MD, Daniel K. Ludwig Distinguished Service Professor and chair of Radiation and Cellular Oncology and a senior author of the study, published in the June 11, 2021 issue of Science Immunology.
ATRA, a form of vitamin A, is thought to play a role in the development and differentiation of immune-related cells. It has been approved by the FDA for use in the treatment of acute promyeloctic leukemia, an aggressive blood cancer, and is considered a more effective and less toxic treatment than chemotherapy.
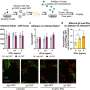
Weichselbaum and a multidisciplinary group of basic science and clinical investigators found the combination of ATRA and radiotherapy produced a much greater antitumor response than that seen with radiotherapy or ATRA alone.
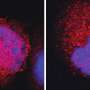
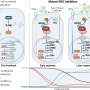
With radiation alone, inflammation in some tumors prompts monocytes (a type of white blood cell) to rush to the scene to repair damage. In the tumor microenvironment, however, some of these monocytes are reprogrammed into myeloid-derived suppressor cells (MDSCs), which promote tumor growth and suppress the tumor-killing power of T cells.
"The addition of ATRA instead creates a positive feedback loop," explained Hua Laura Liang, Ph.D., a Research Assistant Professor at UChicago and co-senior author on the paper. When irradiated, the tumor's pre-existing T cells start producing interferon gamma (IFN-γ), which is important in activating the immune response. Together, IFN-γ and ATRA convert infiltrating monocytes from harmful MDSCs into helpful inflammatory macrophages that produce high levels of two powerful signaling proteins: inducible NO synthase (iNOS) and tumor necrosis factor alpha (TNF-α). These macrophages and the proteins they produce further inflame the tumor microenvironment, which calls in more T cells, starting the cycle anew.
"We can take cells that promote tumor growth and reprogram them to cells that enhance antitumor immunity," Weichselbaum said.
Notably, the combination treatment activated not only CD8+ T cells, which kill cancer cells, but CD4+ T cells, "helper" cells that have a key role in antitumor immunity. Both types of T cells produced elevated levels of IFN-γ, which mediates cancer cell killing.
Of significance was the increased number of both CD8+ and CD4+ T cells in not only locally irradiated tumors but in distal untreated tumors as well, a phenomenon known as the abscopal effect.
Furthermore, the team was able to further suppress the growth of distal tumors by adding to the combination treatment a PD-L1 blockade, an immunotherapy drug that allows T cells to recognize and kill tumor cells. "The addition of a PD-L1 blockade produced a much stronger abscopal effect," Weichselbaum said.
The team sees great potential to use their findings for not only further studies of ATRA-influenced immune cell differentiation but also for bench-to-bedside translation in a clinical trial. "This work has potential clinical significance in using ATRA as a T-cell-targeting compound to improve the local and systemic effects of radiotherapy, thereby benefiting patients with metastatic cancer," Weichselbaum concluded.
More information: All-trans retinoic acid overcomes solid tumor radioresistance by inducing inflammatory macrophages. Science Immunology. DOI: 10.1126/sciimmunol.aba8426