Cancer vaccine could be effective way to overcome treatment resistance
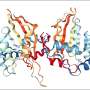
![Fig. 1: CSDE1 is a positive modulator of VSV replication. A Hep3B cells were transfected with no siRNA, Negative control siRNA, or with [s15373 + 15374 siRNA] (2 CSDE1-specific siRNA)46 and levels of CSDE1 assayed by western blotting 24 or 48 h later. (Representative of three separate experiments). B–D Forty-eight hours following transfection with siRNA as in A, Hep3B cells were infected with VSV-GFP (MOI 0.1). Forty-eight hours (B) or 96 h (C) later, viral titers were determined by plaque assay and D the number of surviving cells was counted at 96 h post infection. Representative of two separate experiments. E B16-, B16-CSDE1C-T-, or B16-CSDE1WT-overexpressing cells were infected with VSV-IFN-β at an MOI of 0.1. Twenty-four, 48, and 72 h later, viral titers were measured on BHK cells by plaque assay. Representative of three separate experiments. F Parental Hep3B cells or pooled populations of Hep3B-overexpressing wild-type CSDE1WT, or mutant CSDE1C-T, were infected with VSV-IFN-β (MOI 0.1) (3 wells/group). Forty-eight hours later (Passage 1), supernatants were assayed for infectious titers on the same cells on which the virus was passaged. Virus was recovered every 48 h (P2–5) and similarly titered. Representative of three separate experiments. G Stock VSV-IFN-β virus or VSV-IFN-β, which had been passaged five times through Hep3B parental or Hep3B-CSDE1C-T cells as in F, was titered on either Hep3B parental cells or on Hep3B-CSDE1C-T cells. Representative of two separate experiments. Means ± SD of three technical replicates are shown. P-values were determined using a one-way (B–D) or two-way (E–G) ANOVA with a Tukey’s multiple comparisons post test on log-transformed data. Statistical significance was set at p 0.05. Source data are provided as a Source Data file. Credit: DOI: 10.1038/s41467-021-22115-1 Cancer vaccine could be effective way to overcome treatment resistance](https://scx1.b-cdn.net/csz/news/800a/2021/cancer-vaccine-could-b.jpg)
A cancer vaccine could be an effective way to prevent cancer from evolving and becoming resistant to treatment, new research suggests.
Scientists were investigating the use of a cancer-killing virus in clinical trials, and observed, as they had also seen in mice, that although some patients initially responded to the treatment, their tumors soon became resistant.
The researchers showed that the specific mutations causing tumor cells to become resistant to the viral treatment, could be anticipated and exploited using a vaccine which, when tested in mice, was shown to trigger the immune system to destroy treatment resistant tumor cells.
The findings could be applicable to other types of therapy that drive drug resistance, such as targeted drugs like PARP inhibitors.
A vaccine for cancer evolution
The international study—carried out by scientists at The Institute of Cancer Research, London, the Mayo Clinic in Rochester, US, and the University of Leeds—is the first to show that a vaccine of this type can be used to overcome cancer evolution.
The findings are published in Nature Communications, and the study was funded by The National Institutes of Health, The European Research Council, The Richard M. Schulze Family Foundation, Mayo Foundation, Cancer Research UK, Shannon O'Hara Foundation, and Hyundai Hope on Wheels.
Tumor cells treated with an anti-cancer virus are known to mutate and evolve to escape the virus, rendering the viral treatment less effective.
The scientists found that a predictable mutation occurred in the tumor cells in response to infection with the virus.
Vaccine triggered immune system to destroy cancer cells
Virus treatment caused a mutation within the cancer cells, in a gene called CDSE1, which usually protects against viruses by slowing their replication.
The mutated CDSE1 protein, produced by the CDSE1 gene, created a unique antigen—a structure recognized by the immune system as a target—which the researchers used to develop a vaccine.
The researchers produced a vaccine targeting the mutation, and showed that injecting the vaccine into mice that had been treated with the virus triggered the immune system to destroy the drug resistant tumor cells, killing the cancerous cells and therefore reducing the size of their tumors.
'Trap and ambush' approach
They exploited the tumor's defense mechanism by vaccinating against the mutated cells. This they called a 'trap and ambush' approach; forcing a tumor to evolve in a specific way as a defense mechanism against treatment, and ambushing it by vaccinating against this defense mechanism. This vaccine triggers the immune system to destroy the drug resistant tumor cells, preventing the cancer from further evolving.
This work will be developed further by the new Centre for Translational Immunotherapy (CTI) at The Institute of Cancer Research (ICR), a virtual platform bringing together staff and students from the ICR, and The Royal Marsden NHS Foundation Trust aiming to develop a greater understanding of how immunotherapy works.
Study leader Professor Alan Melcher, Professor of Translational Immunotherapy at The Institute of Cancer Research, London, said:
"In this study, we used a 'trap and ambush' approach, in which initial treatment with a virus caused tumor cells to mutate in a predictable way, before precisely targeting the mutations with a vaccine, preventing the cancer from progressing further.
"This principle may be more widely applicable, as tumors becoming resistant to drugs is a big problem across multiple types of treatment. We are now exploring the possibility of applying this type of approach to other therapies which drive tumor mutation and evolution."
More information: Timothy Kottke et al, Oncolytic virotherapy induced CSDE1 neo-antigenesis restricts VSV replication but can be targeted by immunotherapy, Nature Communications (2021). DOI: 10.1038/s41467-021-22115-1