Study suggests sonic hedgehog protein pathway stimulation could help Parkinson's patients
Levodopa, or L-dopa, is considered the most effective treatment for Parkinson's disease today. After a few years of treatment, however, almost all patients develop a debilitating side-effect called L-dopa induced dyskinesia, or LID, which causes involuntary movements in the limbs, face, and torso. Deep brain stimulation can alleviate LID, but the procedure is highly invasive and not all patients are eligible.
Now, a new study led by researchers at the Graduate Center, CUNY and the CUNY School of Medicine shows that drugs that increased signaling by a protein called sonic hedgehog, or Shh, can inhibit LID. Such a treatment would have the potential to help most Parkinson's patients, the authors said.
The study appears in Communications Biology.
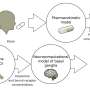
"In rodent and non-human primate models, the administration of L-dopa together with sonic hedgehog agonists attenuate the expression of LID," said Lauren Malave, Ph.D., first author and postdoctoral fellow at Columbia University, previously a Ph.D. student in the lab of Professor Andreas Kottmann, Ph.D., at the CUNY School of Medicine at City College of New York and the Graduate Center. "We provide novel insight into the underlying mechanisms behind LID formation and provide a potential therapeutic solution."
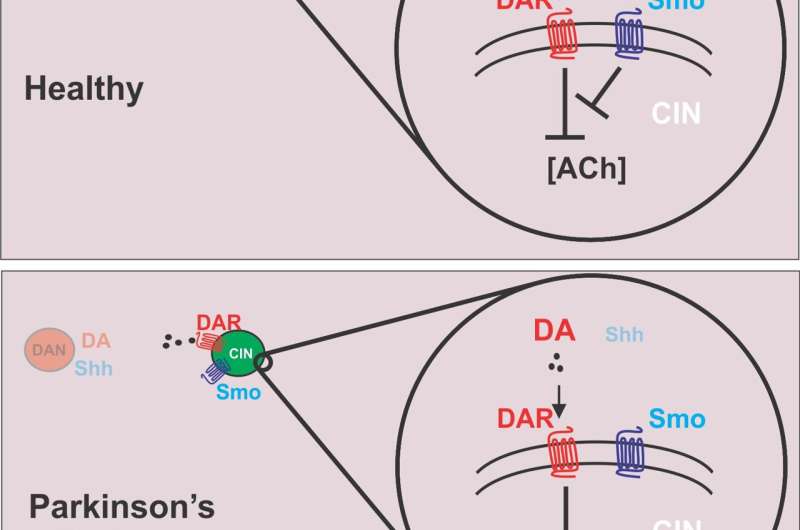
Parkinson's disease is caused by the death of dopamine neurons, which is why the disease is treated with medications that are converted to dopamine once they enter the body. Key to the new study, though, is that these neurons also produce neurotransmitters other than dopamine, including GABA, glutamate, and Shh.
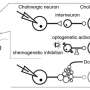
Shh has not previously been considered a neurotransmitter, but the new paper shows that it does in fact act as a neuromodulator. The researchers found that dopamine neurons use Shh to communicate with cholinergic neurons, which scientists have thought might play a role in LID. They then used animal models of Parkinson's disease to show that decreased Shh signaling in the basal ganglia, caused by death of dopamine neurons, facilitates LID. On the other hand, mimicking increased signaling by Shh reduced LID. Because of this, the authors suggest that the imbalance between dopamine and Shh after L-dopa treatment is a major cause of LID.
The next steps will be to develop new therapeutics that act downstream in the Shh pathway in cholinergic neurons and begin clinical trials.
"Deep brain stimulation doesn't help everyone, it's very invasive, and not all people are eligible for the surgery. The procedure is also not accessible to everyone," said Kottmann, who was the corresponding author on the paper. "What we find in this study is that in several animal models, by replacing not only dopamine but dopamine together with agonists that mimic the effects of sonic hedgehog, these dyskinesias can be very much suppressed."
This research was supported by the American Parkinson Disease Association and the National Institutes of Health and the Research Foundation of the City University of New York.
More information: Dopaminergic co-transmission with sonic hedgehog inhibits abnormal involuntary movements in models of Parkinson's disease and L-Dopa induced dyskinesia, Communications Biology (2021). DOI: 10.1038/s42003-021-02567-3