A new drug target for kidney disease
Despite nearly two million people dying of acute kidney injury every year, and tens of millions more needing treatment, no effective drug treatment for the condition is available. Furthermore, most acute kidney injury patients are elderly with poor prognosis, making acute kidney injury a major public health issue and an enormous socioeconomic burden in aging nations. Scientists have therefore searched for unexplored molecular signaling pathways that make promising drug targets. A new study led by ASHBi Professor Motoko Yanagita reports one such pathway by showing a lower expression of two molecules, CD153 and CD30, on specific types of immune cells attenuates dysfunction in aged kidneys.
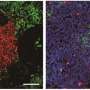
One of the histological hallmarks of acute kidney injury in aged individuals is tertiary lymphoid tissues, which lead to chronic inflammation that can worsen the prognosis. Tertiary lymphoid tissues are also found in populations with several kidney diseases.
Prof. Yanagita explained that tertiary lymphoid tissues provoke an immune reaction involving multiple types of immune cells.
"We assumed that tertiary lymphoid tissues contribute to the progression of kidney injury," she said. "What we don't know is the signaling pathways that govern immune cell interactions."
The immune system responds to infection or injury. However, even in healthy individuals, natural aging affects the function and composition of immune cells, which can lead to a series of diseases commonly seen in the elderly, such as cancer, infections and autoimmunity.
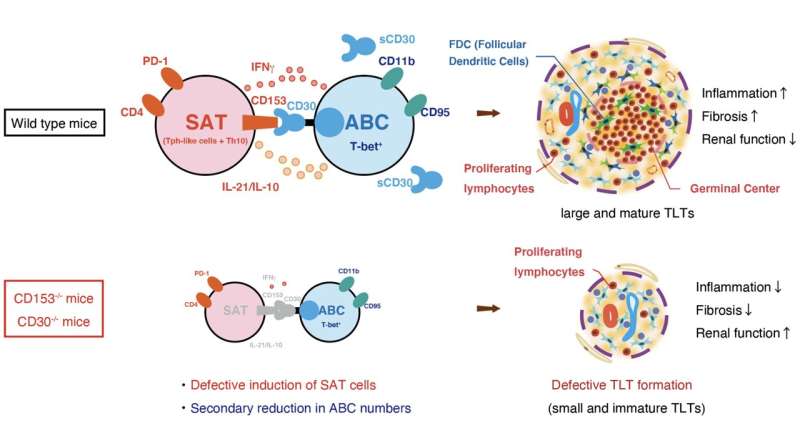
In the study, Yanagita and her colleagues searched for specific immune cells that showed noticeable changes between older mice with healthy and damaged kidneys. They found two specific cell types, senescence-associated T cells (SAT) cells and age-associated B cells (ABCs), continuously accumulated with the formation of tertiary lymphoid tissues in aged mice with injured kidney.
"We found SAT cells and ABCs were linked by TNF superfamily members, CD153 and CD30", remarked Yanagita.
TNF superfamily members are well known in the scientific community for their involvement in the development of lymphoid tissues. CD153 and CD30 are two molecules expressed on the surfaces of SAT cells and ABCs and allow the two cells to communicate with one another. In fact, among the different immune cells examined in the injured mouse kidney, CD153 and CD30 were almost exclusively found on SAT cells and ABCs.
Further, gene knockout of CD153 and CD30 reduced the number of SAT cells and ABCs in mice. More importantly, the knockouts also resulted in fewer tertiary lymphoid tissues and less kidney damage.
An examination of human kidney samples also found that the expression of CD153 and CD30 was higher in immune cells located in tertiary lymphoid tissues.
Noting that tertiary lymphoid tissues are commonly seen with acute kidney injury in mice and humans, Yanagita believes that the findings should be considered for drug discovery.
"The role of CD153 and CD30 in the development of tertiary lymphoid tissues was never appreciated. It could make a promising therapeutic target for kidney treatment in the elderly," she said.
The research was published in the Journal of Clinical Investigation.
More information: Yuki Sato et al, CD153-CD30 signaling promotes age-dependent tertiary lymphoid tissue expansion and kidney injury, Journal of Clinical Investigation (2021). DOI: 10.1172/JCI146071